Chapter 12: Q11-P (page 383)
Where might the following compound have IR absorptions?

Short Answer
Absorption bands seen at 2210-2260 \(c{m^{ - 1}}\), 1690 \(c{m^{ - 1}}\) and 1640-1680 \(c{m^{ - 1}}\).
Learning Materials
Features
Discover
Chapter 12: Q11-P (page 383)
Where might the following compound have IR absorptions?

Absorption bands seen at 2210-2260 \(c{m^{ - 1}}\), 1690 \(c{m^{ - 1}}\) and 1640-1680 \(c{m^{ - 1}}\).
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: At what approximate positions might the following compounds show
IR absorptions?
(c)
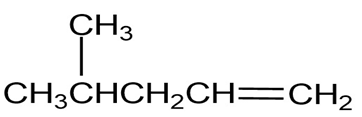
Question: What functional groups might the following molecules contain?
(a) A compound with a strong absorption at 1710
(b) A compound with a strong absorption at 1540
(c) A compound with strong absorptions at 1720 and 2500 to 3100
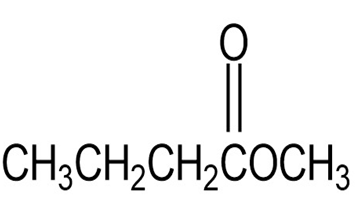
Draw the structure of an alkene that yields only acetone, , on ozonolysis followed by treatment with Zn.
At what approximate positions might the following compounds show
IR absorptions?
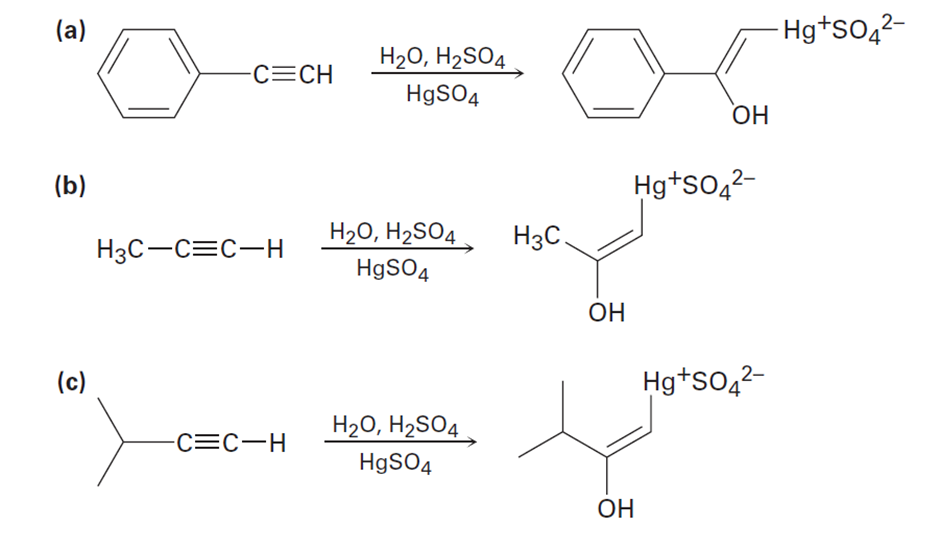
The mercury-catalyzed hydration of alkynes involves the formation of an organomercury enol intermediate. Draw the electron-pushing mechanism to show how each of the following intermediates is formed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.