Chapter 10: Organohalides
10-10-21 E
Alkyl halides can be reduced to alkanes by a radical reaction with tributyltinhydride, in the presence of light (hv). Propose aradical chain mechanism by which the reaction might occur. The initiationstep is the light-induced homolytic cleavage of the Sn-H bondto yield a tributyltin radical.
18E-b
Draw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br∙ and Br2.
(b)
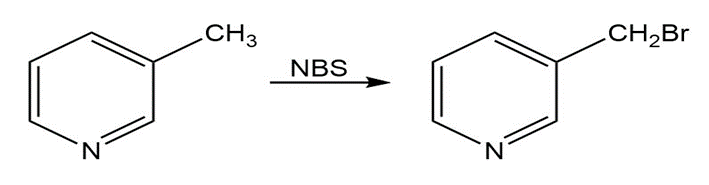
39 E
Question: Assume that you have carried out a radical chlorination reaction on
and have isolated (in low yield) . How many stereoisomers of the product are formed, and in what ratio? Are any of the isomers optically active? (See Problem 10-38.)
42 E
Question: Why do you suppose it’s not possible to prepare a Grignard reagentfrom a bromo alcohol such as ? Give another exampleof a molecule that is unlikely to form a Grignard reagent.
Q10-10P.
Question: How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuterated compound?

Q10-23E-b
Draw structures corresponding to the following IUPAC names: (b) 4-Bromo-4-ethyl-2-methylhexane.
Q10-23E-c
Draw structures corresponding to the following IUPAC names: (c) 3-Iodo-2,2,4,4-tetramethylpentane
Q10-23E-d
Draw structures corresponding to the following IUPAC names: (d) cis-1-Bromo-2-ethylcyclopentane
Q10-25E-a
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (a) Chlorocyclopentane.
Q10-25E-b
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (b) Methylcyclopentane.
