Chapter 10: Q10-10P. (page 300)
Question: How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuterated compound?

Short Answer
Answer
The reaction for the formation of above deuterated compound is as follows:
Learning Materials
Features
Discover
Chapter 10: Q10-10P. (page 300)
Question: How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuterated compound?

Answer
The reaction for the formation of above deuterated compound is as follows:
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (d) Cyclopentanol.
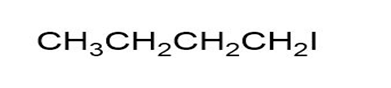
Give IUPAC names for the following alkyl halide (a):
(a)
Tertiary alkyl halides,, undergo spontaneous dissociation to yield a carbocation,, plus halide ion. Which do you think reacts faster,? Explain.
Draw resonance structures for the benzyl radical, the intermediate produced in the NBS bromination reaction of toluene (Problem 10-35).
Draw structures corresponding to the following IUPAC names: (e) 4-sec-Butyl-2-chlorononane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.