Chapter 10: Q10-23E-b (page 308)
Draw structures corresponding to the following IUPAC names: (b) 4-Bromo-4-ethyl-2-methylhexane.
Short Answer
(b)The structure of the compound (b) is given below:

Learning Materials
Features
Discover
Chapter 10: Q10-23E-b (page 308)
Draw structures corresponding to the following IUPAC names: (b) 4-Bromo-4-ethyl-2-methylhexane.
(b)The structure of the compound (b) is given below:

All the tools & learning materials you need for study success - in one app.
Get started for free
The relative rate of radical bromination is 1; 82; 1640 for 1°;2°;3°
hydrogens, respectively. Draw all of the monobrominated products that
you might obtain from the radical bromination of the compounds
below. Calculate the relative percentage of each.
(a)Methylcyclobutane
(b)3,3-dimethylpentane
(c)3-methylpentane
Tell whether each of the following reactions is an oxidation, a reduction, or neither:

Rank the compounds in each of the following series in order of increasing oxidation level:

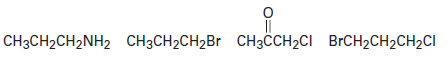
Question: Assume that you have carried out a radical chlorination reaction on
and have isolated (in low yield) . How many stereoisomers of the product are formed, and in what ratio? Are any of the isomers optically active? (See Problem 10-38.)
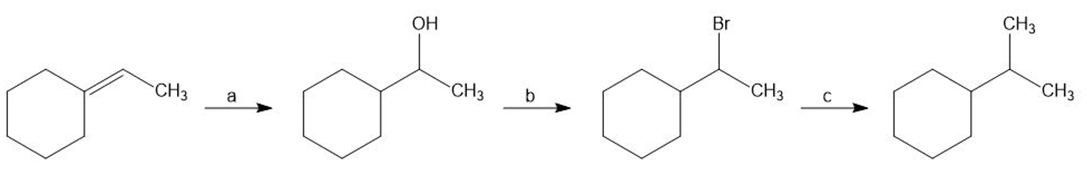
Identify the reagents a–c in the following scheme:
What do you think about this solution?
We value your feedback to improve our textbook solutions.