Chapter 10: Q31 E (page 308)
Rank the compounds in each of the following series in order of increasing oxidation level:
Learning Materials
Features
Discover
Chapter 10: Q31 E (page 308)
Rank the compounds in each of the following series in order of increasing oxidation level:
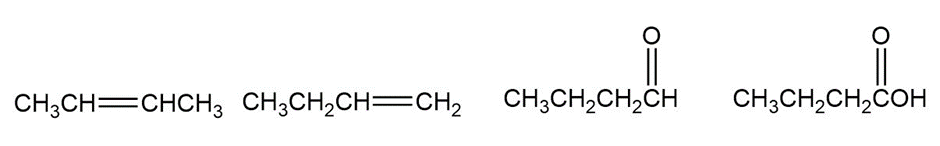

All the tools & learning materials you need for study success - in one app.
Get started for free
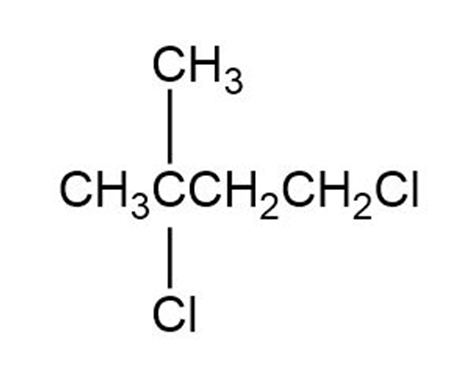
Give IUPAC names for the following alkyl halide (d):
Question: The syntheses shown here are unlikely to occur as written. What is
wrong with each?
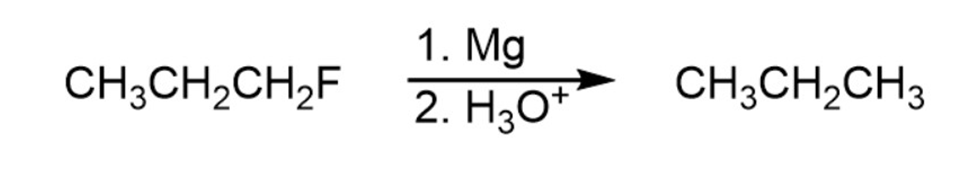
Reaction 1
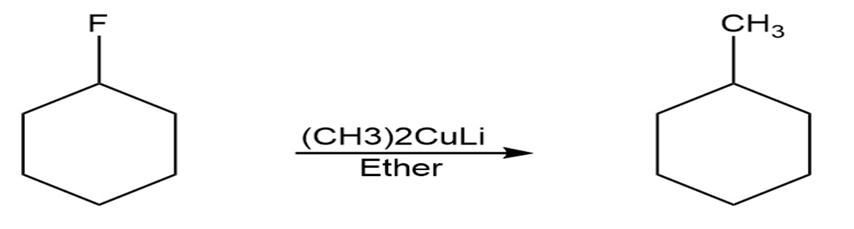
Reaction 2

Draw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br∙ and
(c)
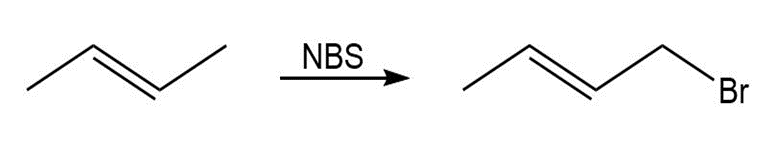
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (c) 3-Bromocyclopentene.
Question: Assume that you have carried out a radical chlorination reaction on
and have isolated (in low yield) . How many stereoisomers of the product are formed, and in what ratio? Are any of the isomers optically active? (See Problem 10-38.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.