Chapter 16: Chemistry of Benzene: Electrophilic Aromatic Substitution
Q10 P
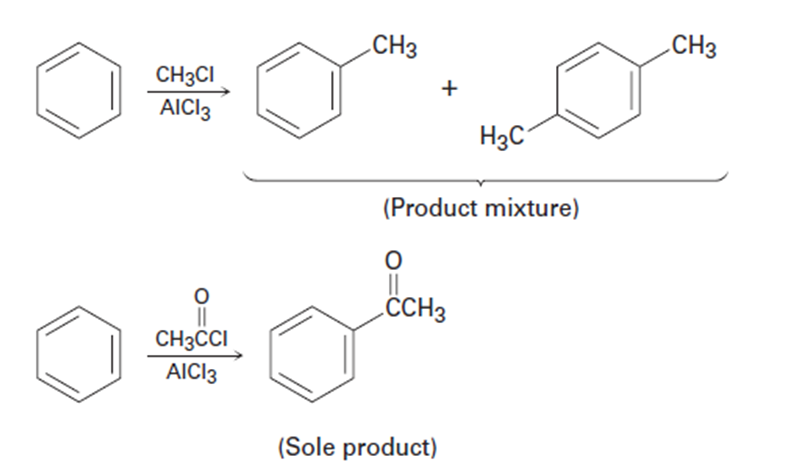
Use Figure 16-11 to explain why Friedel–Crafts alkylations often give polysubstitution but Friedel–Crafts acylations do not.
Q11 P
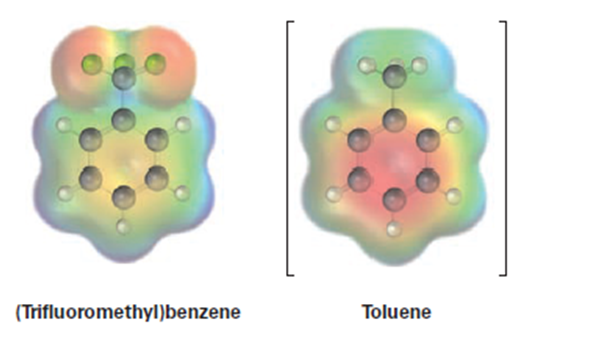
An electrostatic potential map of (trifluoromethyl)benzene, , is shown. Would you expect (trifluoromethyl)benzene to be more reactive orless reactive than toluene toward electrophilic substitution? Explain.
Q12 P
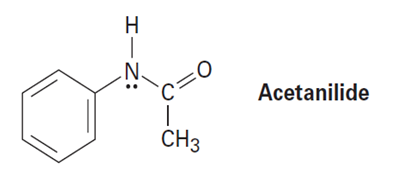
Acetanilide is less reactive than aniline toward electrophilic substitution. Explain
Q13 P
Draw resonance structures for the intermediates from the reaction of an electrophile at the ortho, meta, and para positions of nitrobenzene. Which intermediates are most stable?
Q14 P
At what position would you expect electrophilic substitution to occur in each of the following substances?
a.
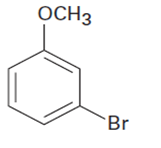
b.
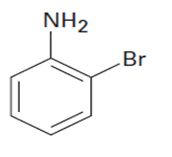
c.
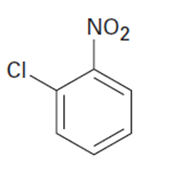
Q16-15P
Show the major product(s) from reaction of the following substances with
(1) , and
(2) , :
a.
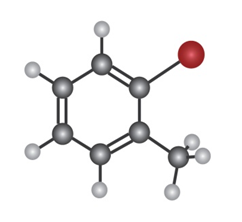
b.
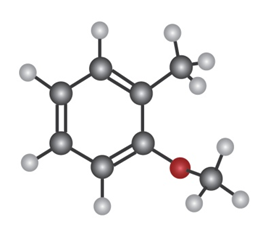
Q16-16P
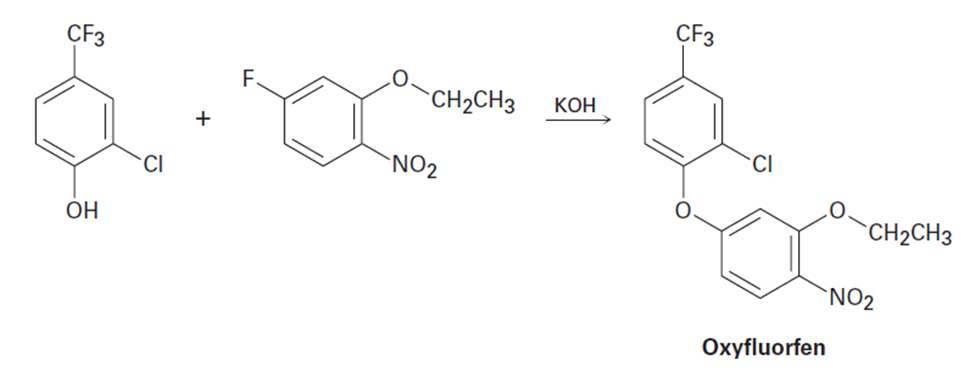
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose a mechanism.
Q16-17P
Treatment of p-bromotoluene with NaOH at yields a mixture of twoproducts, but treatment of m-bromotoluene with NaOH yields a mixture ofthreeproducts. Explain.
Q16-18P
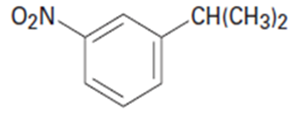
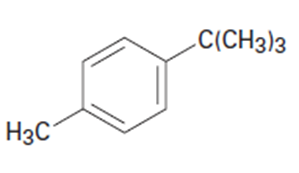
What aromatic products would you obtain from the oxidation of the following substances?
a.
b.
Q16-19P
Refer to Table 6-3 on page 170 for a quantitative idea of the stability of a benzyl radical. How much more stable (in kJ/mol) is the benzyl radical than a primary alkyl radical? How does a benzyl radical compare in stability to an allylradical?
