Chapter 16: Q13 P (page 502)
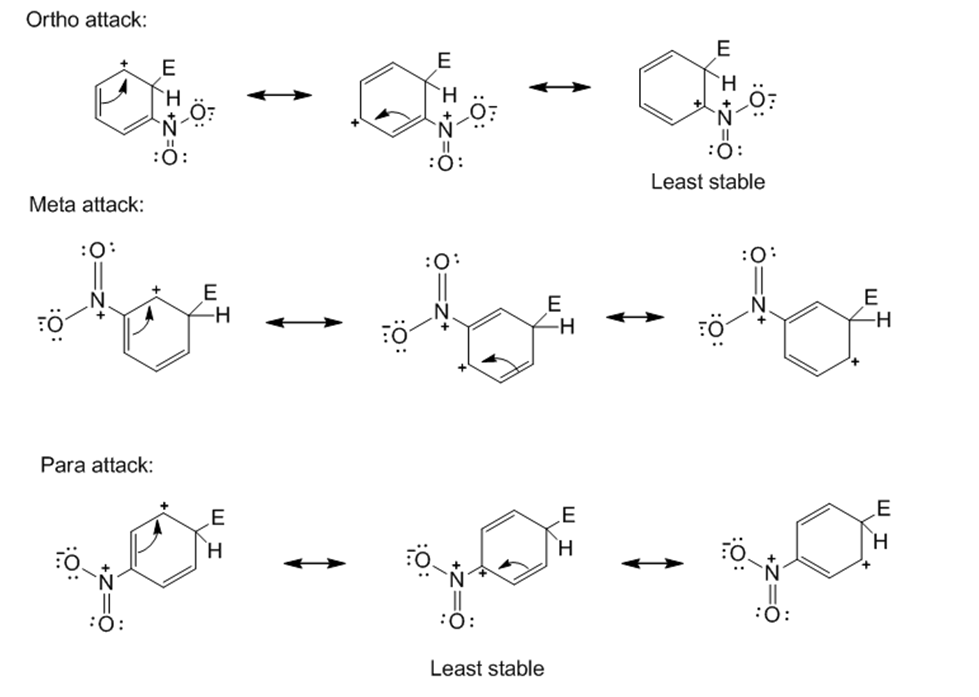
Draw resonance structures for the intermediates from the reaction of an electrophile at the ortho, meta, and para positions of nitrobenzene. Which intermediates are most stable?
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q13 P (page 502)
Draw resonance structures for the intermediates from the reaction of an electrophile at the ortho, meta, and para positions of nitrobenzene. Which intermediates are most stable?

All the tools & learning materials you need for study success - in one app.
Get started for free
The sulfonation of an aromatic ring with is reversible. That is, heating benzenesulfonic acid with H2SO4 yields benzene. Show the mechanism of the desulfonation reaction. What is the electrophile?
Predict the major product (s) you would obtain from sulfonation of the following compounds:
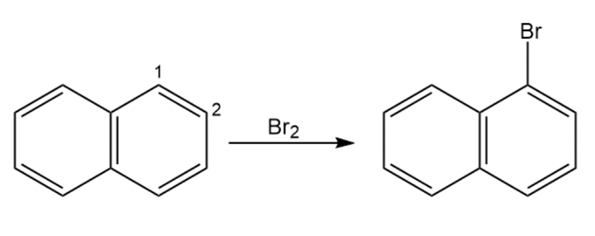
Draw resonance structures of the intermediate carbocations in the bromination of naphthalene, and account for the fact that naphthalene undergoes electrophilic substitution at C1 rather than C2.
Styrene, the simplest alkenylbenzene, is prepared commercially for use in plastics manufacture by catalytic dehydrogenation of ethylbenzene. Howmight you prepare styrene from benzene using reactions you’ve studied?
At what position would you expect electrophilic substitution to occur in each of the following substances?
a.
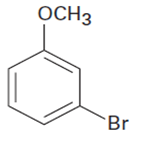
b.
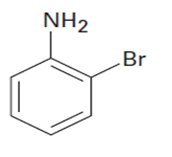
c.
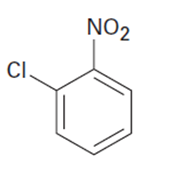
What do you think about this solution?
We value your feedback to improve our textbook solutions.