Chapter 1: Structure and Bonding
48E
Allene (see Problem 1-47) is structurally related to carbon dioxide, . Draw a picture showing the orbitals involved in the s and p bonds of , and identify the likely hybridization of carbon.
Q10P
Draw a line-bond structure for propene, . Indicate the hybridization of the orbitals on each carbon, and predict the value of each bond angle.
Q1-1-19P
The following model is a representation of citric acid, the key substance in the so-called citric acid cycle, by which food molecules are metabolized in the body. Only the connections between atoms are shown; multiple bonds are not indicated. Complete the structure by indicating the positions of multiple bonds and lone-pair electrons (gray 5 C, red 5 O, ivory 5 H).
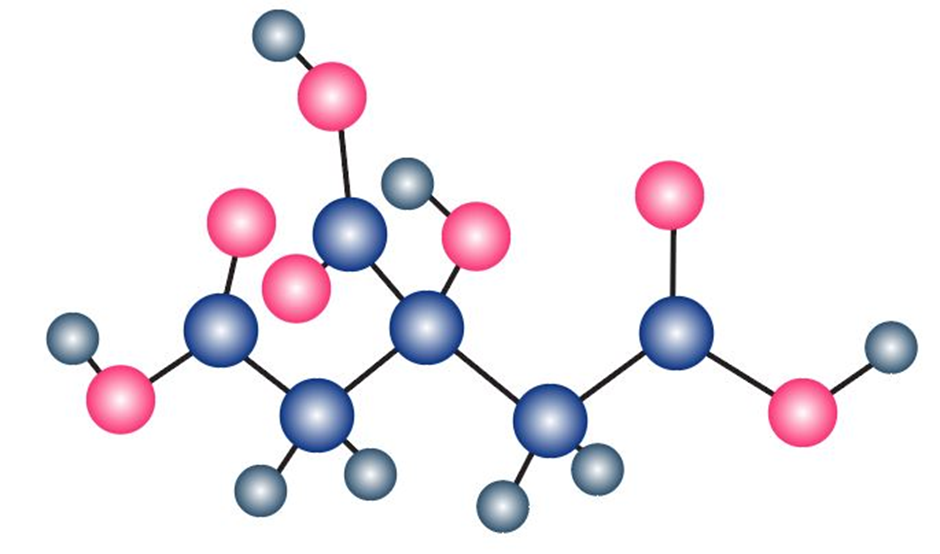
Q1-1-1P
Give the ground-state electron configuration for each of the following elements:
(a) Oxygen (b) Nitrogen (c) Sulfur
Q-1-1-22E
How many valence electrons does each of the following dietary trace elements have?
(a) Zinc (b) Iodine (c) Silicon (d) Iron
Q.1-1-23a
Give the ground-state electron configuration for each of the following elements:
(a)Potassium (b) Arsenic (c) Aluminum (d) Germanium
Q.1-1-23b
Give the ground-state electron configuration for each of the following elements:
(a) Potassium (b) Arsenic (c) Aluminum (d) Germanium
(b)
Q.1-1-23c
Give the ground-state electron configuration for each of the following elements:
(a) Potassium (b) Arsenic (c) Aluminum (d) Germanium
(c)
Q.1-1-23d
Give the ground-state electron configuration for each of the following elements:
(a) Potassium (b) Arsenic (c) Aluminum (d) Germanium
(d)
Q.1-1-24a
What are likely formulas for the following molecules?
(a) NH?OH (b)AlCl? (c) CF2Cl?(d)CHO?
(a)
