Chapter 1: Structure and Bonding
Q.1-1-24b
What are likely formulas for the following molecules?
(a) NH?OH (b) AlCl?(c) CF2Cl(d)CHO?
(b)
Q.1-1-24d
What are likely formulas for the following molecules?
(a) (b) (c) (d)
Q.1-1-25a
Why can’t molecules with the following formulas exist
a) (b) (c)
Q.1-1-25b
Why can’t molecules with the following formulas exist
a) (b)(c)
Q.1-1-25c
Why can’t molecules with the following formulas exist
a) (b) (c)
Q 1-1-26E
Draw an electron-dot structure for acetonitrile,, which contains a carbon-nitrogen triple bond. How many electrons does the nitrogen atom have in its outer shell? How many are bonding, and how many are nonbonding?
Q1-1-27E
Draw a line-bond structure for vinyl chloride,C2H3Cl, the starting material from which PVC [poly(vinyl chloride)] plastic is made
Q. 1-1-28E
Fill in any nonbonding valence electrons that are missing from the following structures
(a)
 Dimethyl sulfide
Dimethyl sulfide
(b)
 Acetamide
Acetamide
(c)
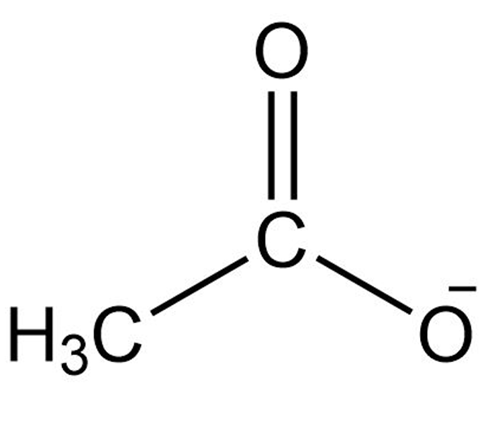
Acetate ion
Q. 1-1-29E
Convert the following line-bond structures into molecular formulas
(a)
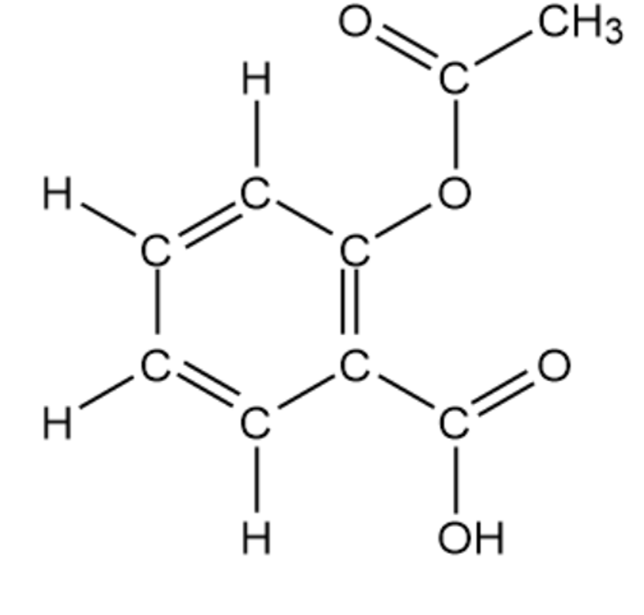
Aspirin (acetyl salicylic acid)
(b)
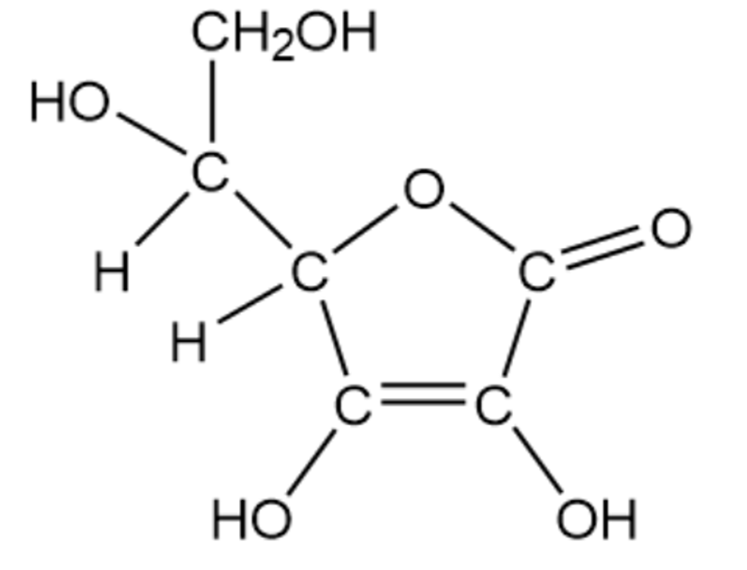
Vitamin C (ascorbic acid)
(c)
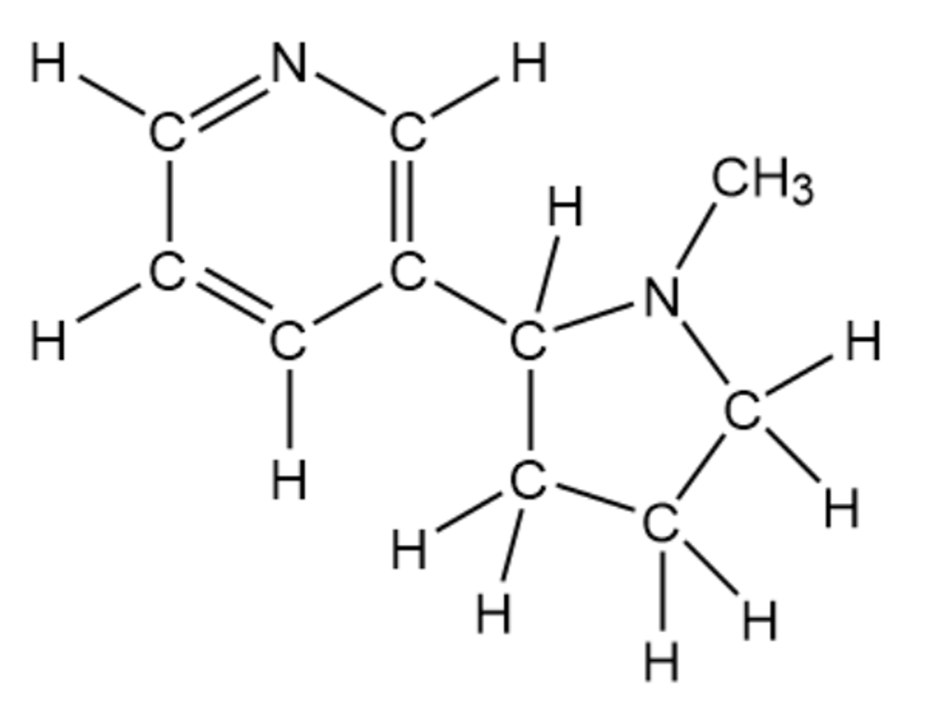
Nicotine
(d)
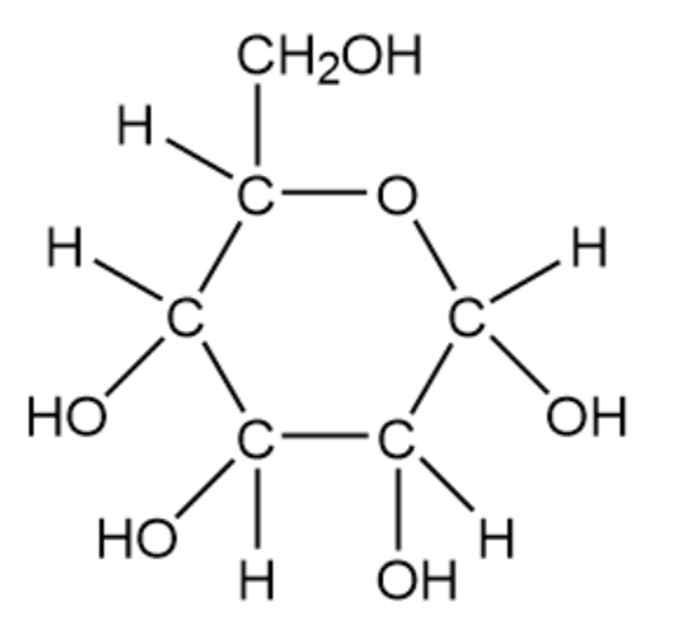
Glucose
Q1-12P
Following is a molecular model of aspirin (acetylsalicylic acid). Identify the hybridization of the orbitals on each carbon atom in aspirin, and tell which atoms have lone pairs of electrons (gray 5 C, pink 5 O, ivory 5 H).

Aspirin (acetylsalicylic acid)
