Chapter 1: Q.1-1-25b (page 27)
Why can’t molecules with the following formulas exist
a) (b)(c)
Short Answer
Answer
The formula can’t exist.
Learning Materials
Features
Discover
Chapter 1: Q.1-1-25b (page 27)
Why can’t molecules with the following formulas exist
a) (b)(c)
Answer
The formula can’t exist.
All the tools & learning materials you need for study success - in one app.
Get started for free
What are likely formulas for the following molecules?
(a) (b) (c) (d)
Propose structures for molecules that meet the following descriptions:
(a) Contains two sp2 -hybridized carbons and two -hybridized carbons
(b) Contains only four carbons, all of which are -hybridized
(c) Contains two -hybridized carbons and two -hybridized carbons
Potassium methoxide,,KOCH3 contains both covalent and ionic bonds. Which do you think is which?
Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers such ibuprofen (Advil, Motrin), naproxen (Aleve), and acetaminophen (Tylenol).
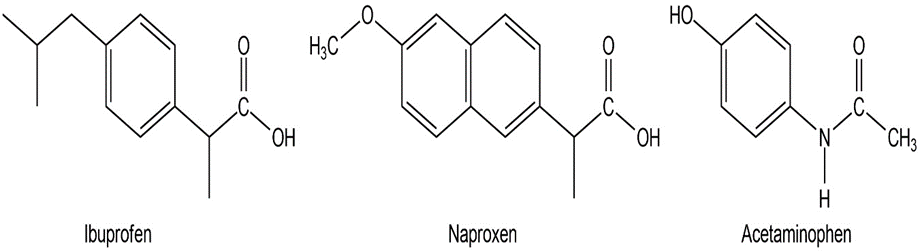
(a) How many sp3-hybridized carbons does each molecule have?
(b) How many sp2-hybridized carbons does each molecule have?
(c) Can you spot any similarities in their structures?
What is the hybridization of each carbon atom in acetonitrile?
What do you think about this solution?
We value your feedback to improve our textbook solutions.