Chapter 1: Q.1-1-25c (page 27)
Why can’t molecules with the following formulas exist
a) (b) (c)
Short Answer
Answer
The formula can’t exist.
Learning Materials
Features
Discover
Chapter 1: Q.1-1-25c (page 27)
Why can’t molecules with the following formulas exist
a) (b) (c)
Answer
The formula can’t exist.
All the tools & learning materials you need for study success - in one app.
Get started for free
Convert the following structures into skeletal drawings
(a)
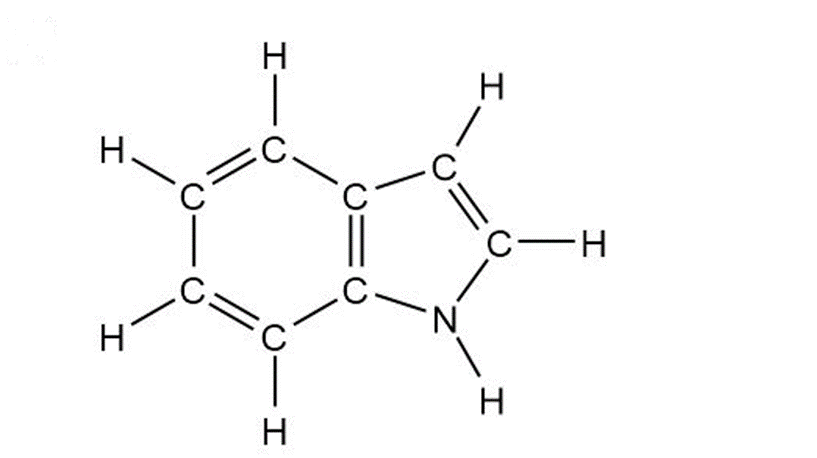
Indole
Identify all nonbonding lone pairs of electrons in the following molecules, and tell what geometry you expect for each of the indicated atoms.
Draw a line-bond structure for propane, . Predict the value of each bond angle, and indicate the overall shape of the molecule.
Allene (see Problem 1-47) is structurally related to carbon dioxide, . Draw a picture showing the orbitals involved in the s and p bonds of , and identify the likely hybridization of carbon.
Draw a line-bond structure for vinyl chloride,C2H3Cl, the starting material from which PVC [poly(vinyl chloride)] plastic is made
What do you think about this solution?
We value your feedback to improve our textbook solutions.