Chapter 1: Q8P (page 14)
Draw a line-bond structure for propane, . Predict the value of each bond angle, and indicate the overall shape of the molecule.
Short Answer
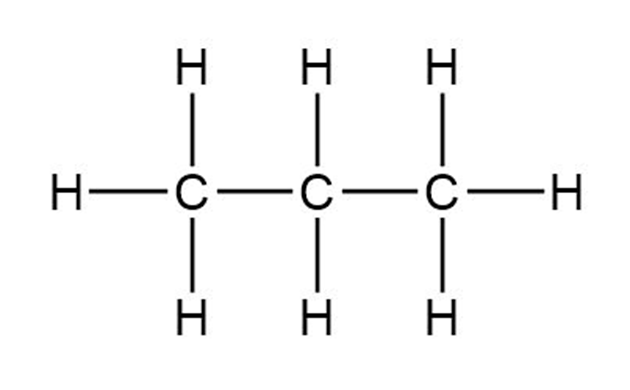
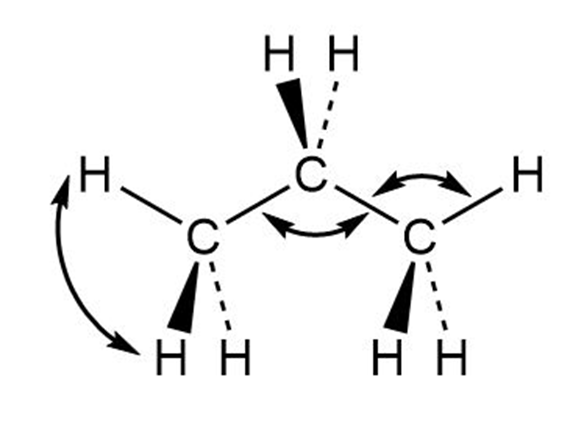
Each bond angle is
Learning Materials
Features
Discover
Chapter 1: Q8P (page 14)
Draw a line-bond structure for propane, . Predict the value of each bond angle, and indicate the overall shape of the molecule.


Each bond angle is
All the tools & learning materials you need for study success - in one app.
Get started for free
How many electrons does each of the following elements have in its outermost electron shell?
a) Magnesium (b) Cobalt (c) Selenium
What are likely formulas for the following molecules?
(a) (b) (c) (d)
What are likely formulas for the following molecules?
(a) (b) (c) (d)
A carbanion is a species that contains a negatively charged, trivalent carbon.
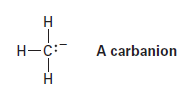
(a) What is the electronic relationship between a carbanion and a trivalent nitrogen compound such as NH3?
(b) How many valence electrons does the negatively charged carbon atom have?
(c) What hybridization do you expect this carbon atom to have?
(d) What geometry is the carbanion likely to have?
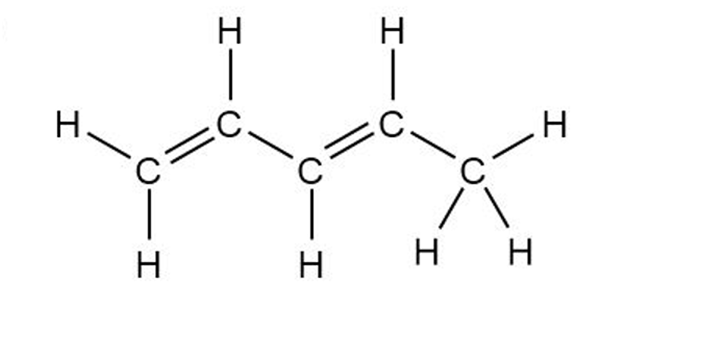
Convert the following structures into skeletal drawings
(a)
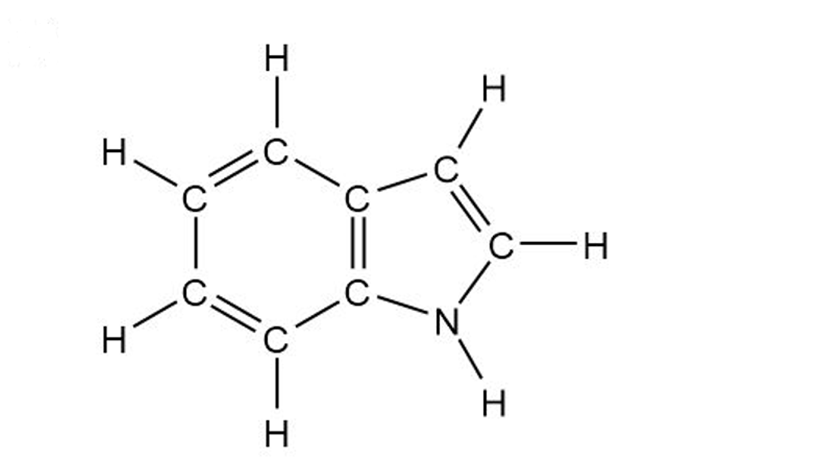
Indole
(b)
Quetiapine(Seroquel)
1,3-pentadiene
(c)
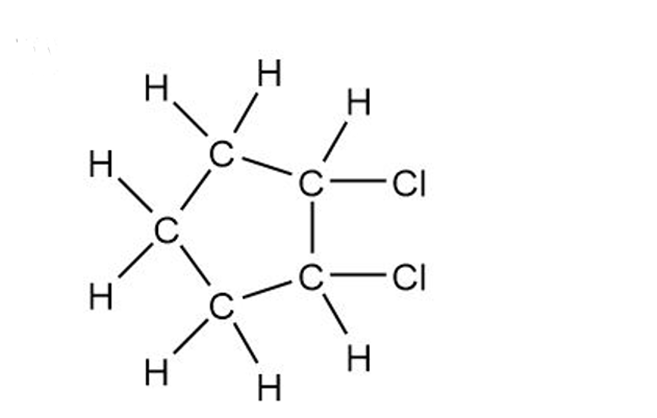
1,2-dicholorcyclopentane
(d)
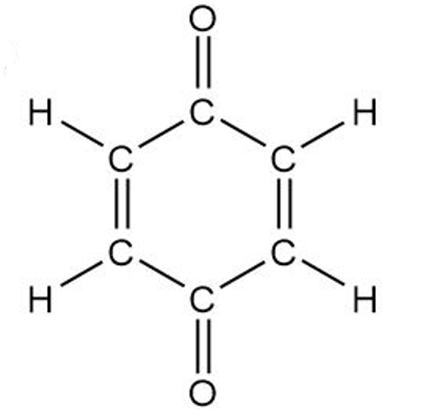
Benzoquinone
What do you think about this solution?
We value your feedback to improve our textbook solutions.