Chapter 8: Reactions and Synthesis
52aE
Show the structures of alkenes that give the following products on oxidative cleavage within acidic solution:
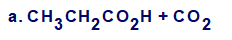
8-31
Propose a curved-arrow mechanism to show how ozone (O3) reacts
with a carbon–carbon double bond to form a molozonide, the first intermediate
in ozonolysis.
8-32a
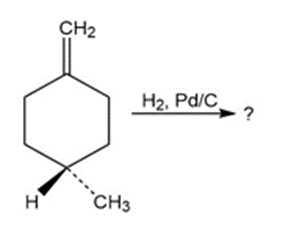
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
a)
8-32b
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
b)
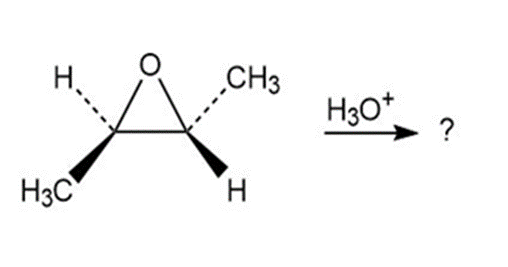
8-32c
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
c)
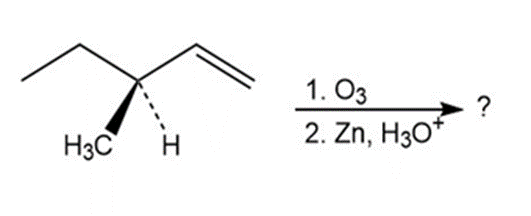
8-34a
Iodine azide, adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as 1-butene isused, only one product results:
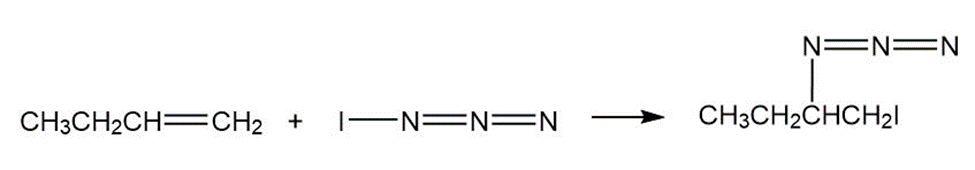
a) Add lone-pair electrons to the structure shown for, and draw a
second resonance form for the molecule.
8-34b
Iodine azide,adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as1-butene isused, only one product results:
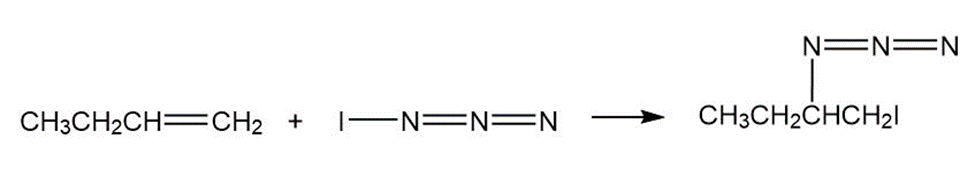
b) Calculate formal charges for the atoms in both resonance structuresyou drew for,in part (a).
8-34c
Iodine azide,, adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as 1-butene is used, only one product results:
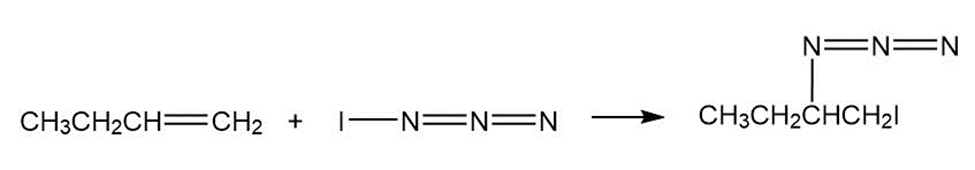
c)
In light of the result observed when,adds to 1-butene, what is
the polarity of thebond? Propose a mechanism for the reaction
using curved arrows to show the electron flow in each step.
8-35
10-Bromo-a-chamigrene, a compound isolated from marine algae, isthought to be biosynthesized from g-bisabolene by the following route:
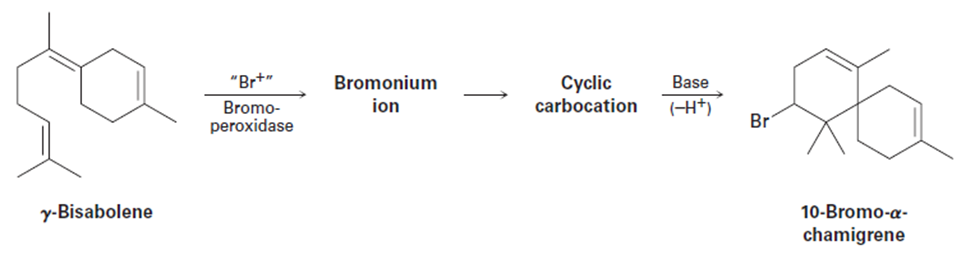
Draw the structures of the intermediate bromonium and cyclic carbocation,and propose mechanisms for all three steps.
8-36
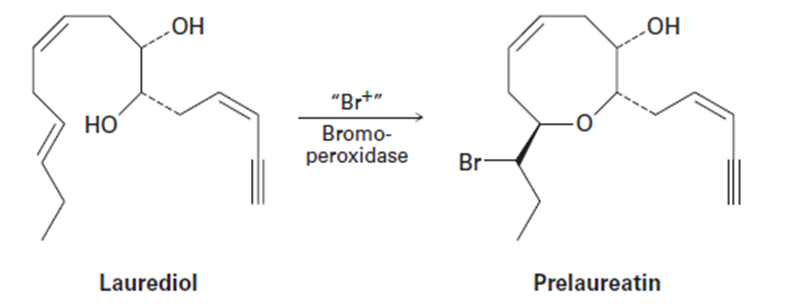
Isolated from marine algae, prelaureatin is thought to be biosynthesized from laurediol by the following route. Propose a mechanism