Chapter 8: 8-36 (page 262)
Isolated from marine algae, prelaureatin is thought to be biosynthesized from laurediol by the following route. Propose a mechanism
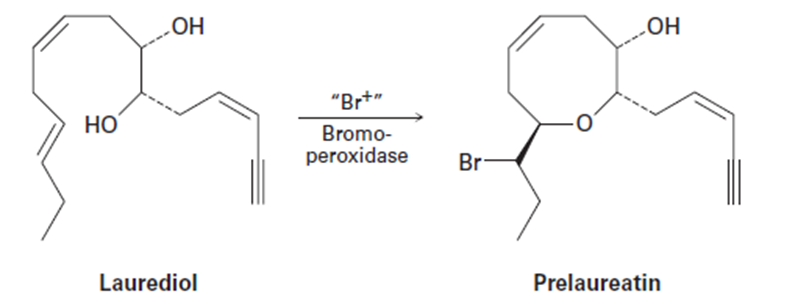
Short Answer
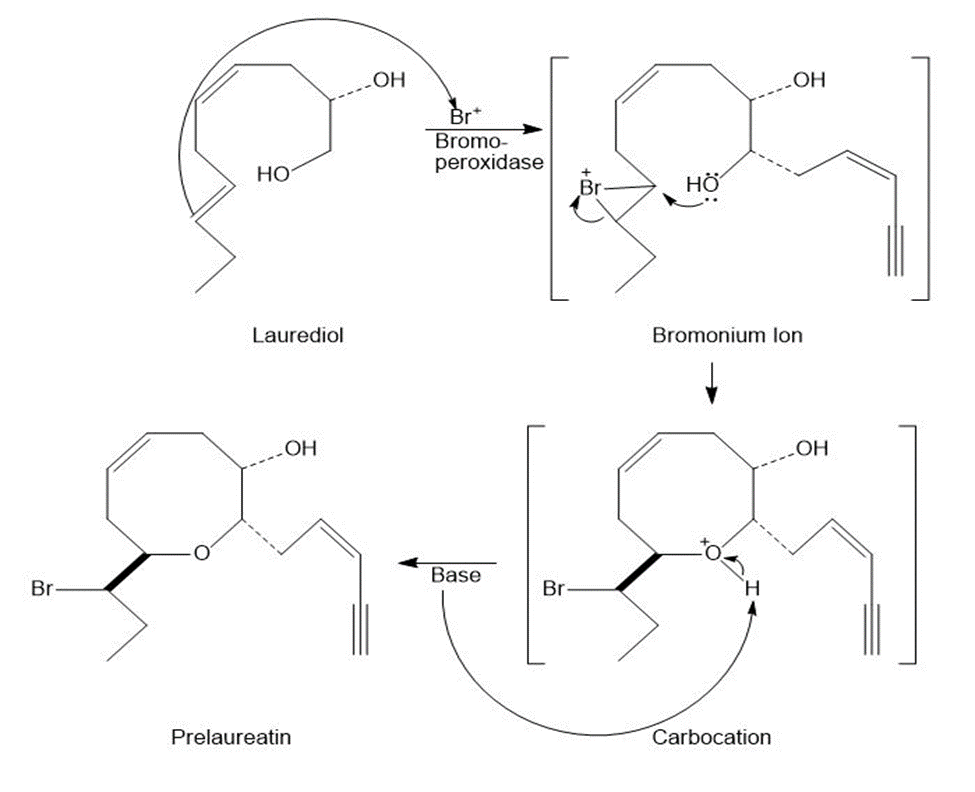
Mechanism of the reaction is:
Learning Materials
Features
Discover
Chapter 8: 8-36 (page 262)
Isolated from marine algae, prelaureatin is thought to be biosynthesized from laurediol by the following route. Propose a mechanism

Mechanism of the reaction is:

All the tools & learning materials you need for study success - in one app.
Get started for free
Simmons–Smith reaction of cyclohexene with diiodomethane gives asingle cyclopropane product, but the analogous reaction of cyclohexene with 1,1-diiodoethane gives (in low yield) a mixture of two isomeric methylcyclopropane products. What are the two products, and how do they differ?
Iodine azide,, adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as 1-butene is used, only one product results:
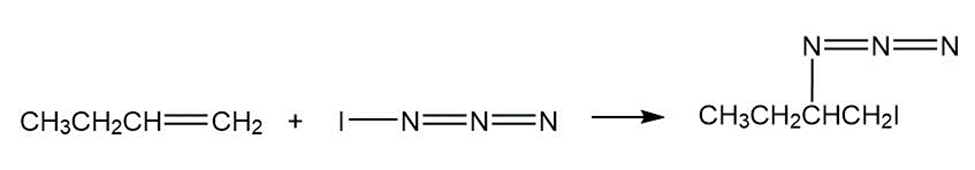
c)
In light of the result observed when,adds to 1-butene, what is
the polarity of thebond? Propose a mechanism for the reaction
using curved arrows to show the electron flow in each step.
The reaction of cyclohexene with mercury(II) acetate in CH3OH rather than
H2O, followed by treatment with NaBH4, yields cyclohexyl methyl ether rather than cyclohexanol. Suggest a mechanism.
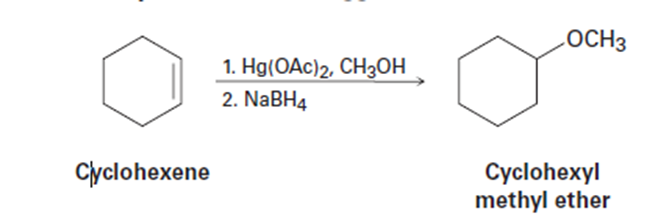
Question: In planning the synthesis of one compound from another, it’s just as important to know what not to do as to know what to do. The following reactions all have serious drawbacks to them. Explain the potential problems of each.
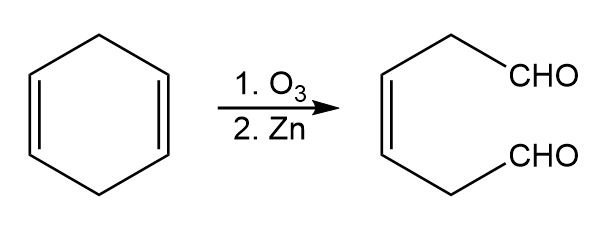
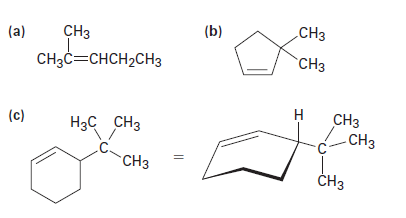
What products would you obtain from catalytic hydrogenation of the following alkenes?
What do you think about this solution?
We value your feedback to improve our textbook solutions.