Chapter 8: 8-5 (page 225)
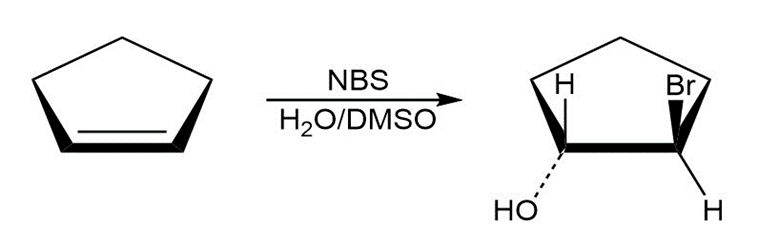
What product would you expect from the reaction of cyclopentene with NBS and water? Show the stereochemistry.
Short Answer
The product formed by the reaction of cyclopentene with NBS and water is,
Learning Materials
Features
Discover
Chapter 8: 8-5 (page 225)
What product would you expect from the reaction of cyclopentene with NBS and water? Show the stereochemistry.
The product formed by the reaction of cyclopentene with NBS and water is,

All the tools & learning materials you need for study success - in one app.
Get started for free
Iodine azide,adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as1-butene isused, only one product results:
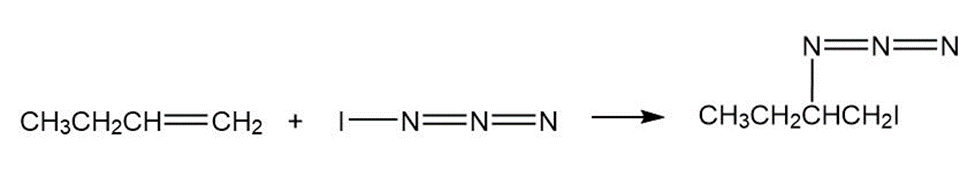
b) Calculate formal charges for the atoms in both resonance structuresyou drew for,in part (a).
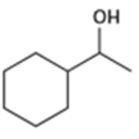
From what alkenes might the following alcohols have been prepared?
b)
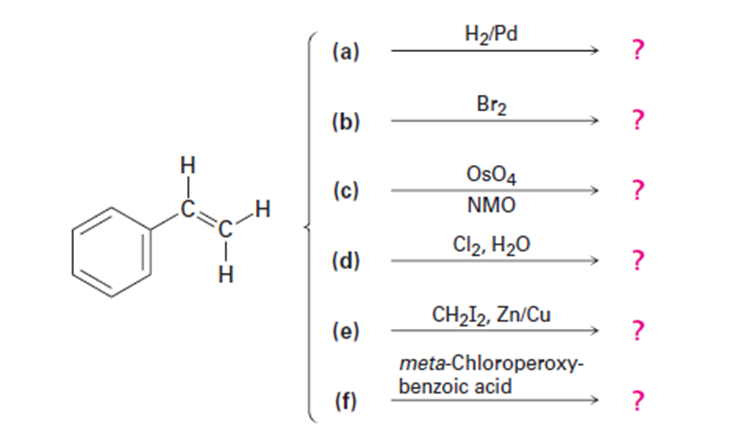
Predict the products of the following reactions (the aromatic ring is
unreactive in all cases). Indicate regiochemistry when relevant.
Iodine azide,, adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as 1-butene is used, only one product results:
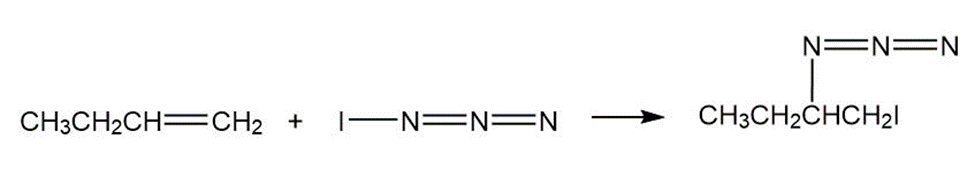
c)
In light of the result observed when,adds to 1-butene, what is
the polarity of thebond? Propose a mechanism for the reaction
using curved arrows to show the electron flow in each step.
Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution
What do you think about this solution?
We value your feedback to improve our textbook solutions.