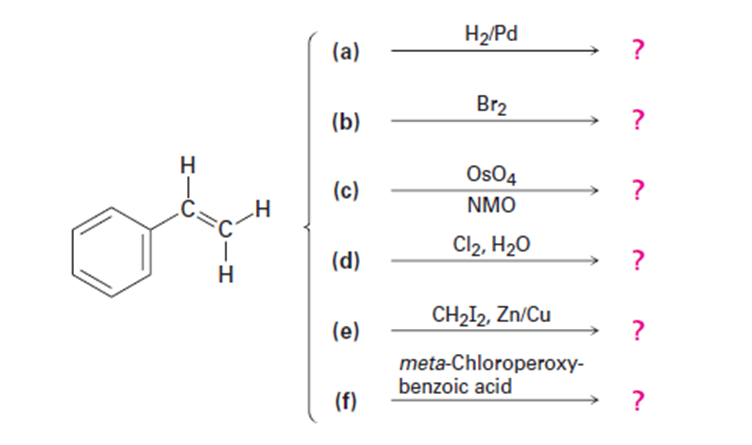
Chapter 8: Q42E (page 220)
Predict the products of the following reactions (the aromatic ring is
unreactive in all cases). Indicate regiochemistry when relevant.
Short Answer
The product of the following reactions is as shown below:
Learning Materials
Features
Discover
Chapter 8: Q42E (page 220)
Predict the products of the following reactions (the aromatic ring is
unreactive in all cases). Indicate regiochemistry when relevant.

The product of the following reactions is as shown below:
All the tools & learning materials you need for study success - in one app.
Get started for free
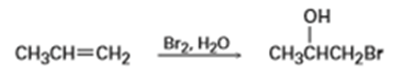
When an unsymmetrical alkene such as propene is treated with N-bromosuccinimide in aqueous dimethyl sulfoxide, the major product has the bromine atom bonded to the less highly substituted carbon atom. Is this Markovnikov or non-Markovnikov orientation? Explain
We’ll see in the next chapter that alkynes undergo many of the same reactions that alkenes do. What product might you expect from each of the following reactions?
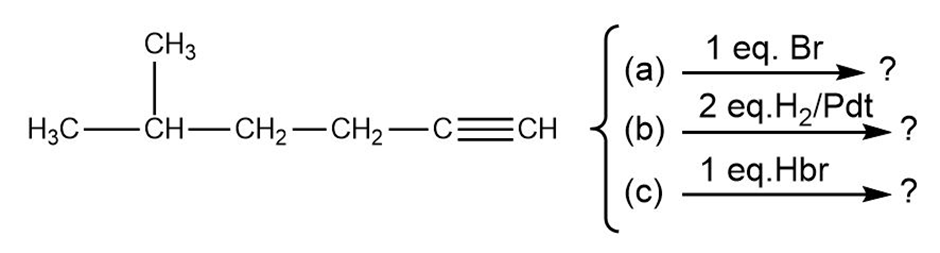
Propose a curved-arrow mechanism to show how ozone (O3) reacts
with a carbon–carbon double bond to form a molozonide, the first intermediate
in ozonolysis.
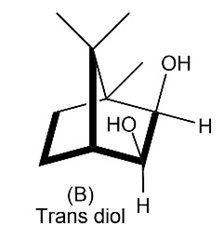
Evidence that cleavage of 1,2-diols by occurs through a five membered cyclic periodate intermediate is based on kinetic data—the measurement of reaction rates. When diols A and B were prepared and the rates of their reaction withwere measured, it was found that diol A cleaved approximately 1 million times faster than diol B. Make molecular models of A and B and of potential cyclic periodate intermediates, and then explain the kinetic results

Iodine azide,, adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as 1-butene is used, only one product results:
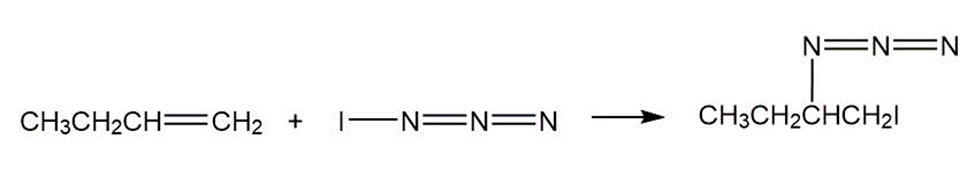
c)
In light of the result observed when,adds to 1-butene, what is
the polarity of thebond? Propose a mechanism for the reaction
using curved arrows to show the electron flow in each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.