Chapter 8: Q39E (page 220)
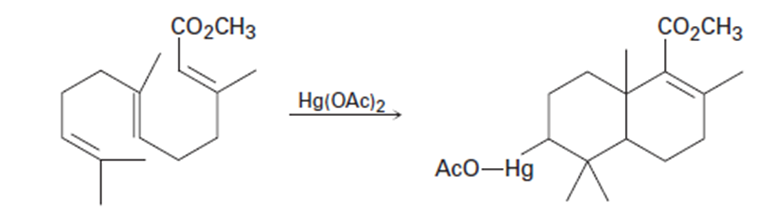
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction.
Learning Materials
Features
Discover
Chapter 8: Q39E (page 220)
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction.

All the tools & learning materials you need for study success - in one app.
Get started for free
Simmons–Smith reaction of cyclohexene with diiodomethane gives asingle cyclopropane product, but the analogous reaction of cyclohexene with 1,1-diiodoethane gives (in low yield) a mixture of two isomeric methylcyclopropane products. What are the two products, and how do they differ?
How would you distinguish between the following pairs of compounds using simple chemical tests? Tell what you would do and what you would see.
(b) 2-Hexene and benzene
Compound A has the formula . It reacts rapidly with to give and a carboxylic acid, B , but reacts with only 1 molar equivalent of on catalytic hydrogenation over a palladium catalyst. On hydrogenation under conditions that reduce aromatic rings, 4 equivalents of are taken up and hydrocarbon C is produced. What are the structures of A, B, and C? Write the reactions.
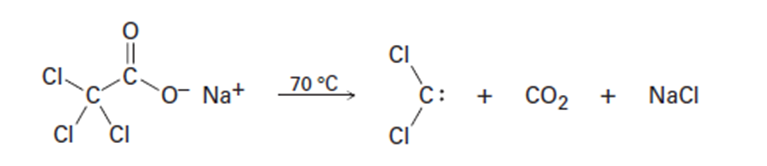
Dichlorocarbene can be generated by heating sodium trichloroacetate.
Propose a mechanism for the reaction, and use curved arrows to indicate
the movement of electrons in each step. What relationship does
your mechanism bear to the base-induced elimination of HCl from
chloroform?
The reaction of cyclohexene with mercury(II) acetate in CH3OH rather than
H2O, followed by treatment with NaBH4, yields cyclohexyl methyl ether rather than cyclohexanol. Suggest a mechanism.
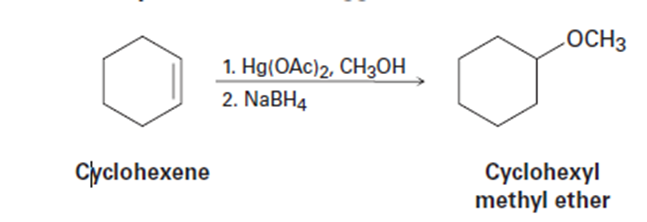
What do you think about this solution?
We value your feedback to improve our textbook solutions.