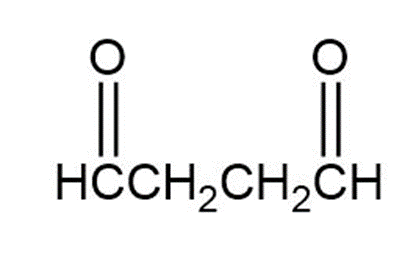
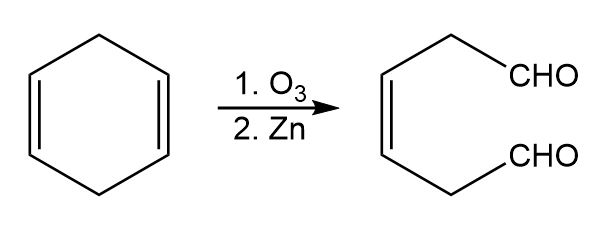
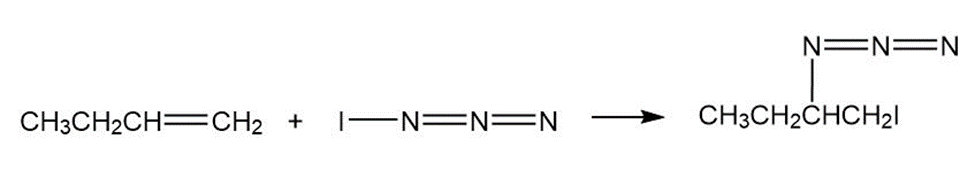Chapter 8: Q8-63 E (page 262)
Simmons–Smith reaction of cyclohexene with diiodomethane gives asingle cyclopropane product, but the analogous reaction of cyclohexene with 1,1-diiodoethane gives (in low yield) a mixture of two isomeric methylcyclopropane products. What are the two products, and how do they differ?
Short Answer
Answer
Enantiomers, have R and S configurations.