Chapter 8: 8-31 (page 262)
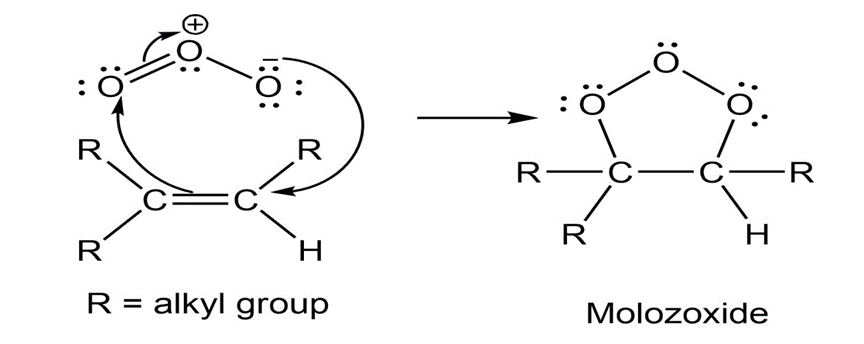
Propose a curved-arrow mechanism to show how ozone (O3) reacts
with a carbon–carbon double bond to form a molozonide, the first intermediate
in ozonolysis.
Short Answer
Curved-arrow mechanism:
Learning Materials
Features
Discover
Chapter 8: 8-31 (page 262)
Propose a curved-arrow mechanism to show how ozone (O3) reacts
with a carbon–carbon double bond to form a molozonide, the first intermediate
in ozonolysis.
Curved-arrow mechanism:

All the tools & learning materials you need for study success - in one app.
Get started for free
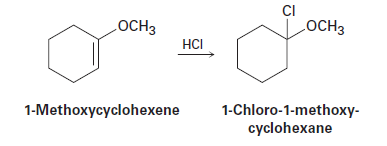
Question:The addition of HCl to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclohexane as a sole product. Use resonance structures of the carbocation intermediate to explain why none of the alternate regioisomers isformed.
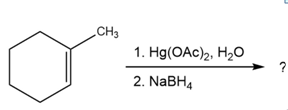
Question:Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate:
(d)
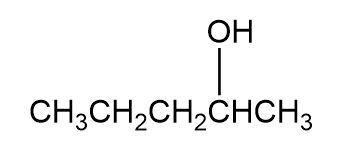
Question: Which of the following alcohols could notbe made selectively by hydroboration–oxidation of an alkene? Explain.
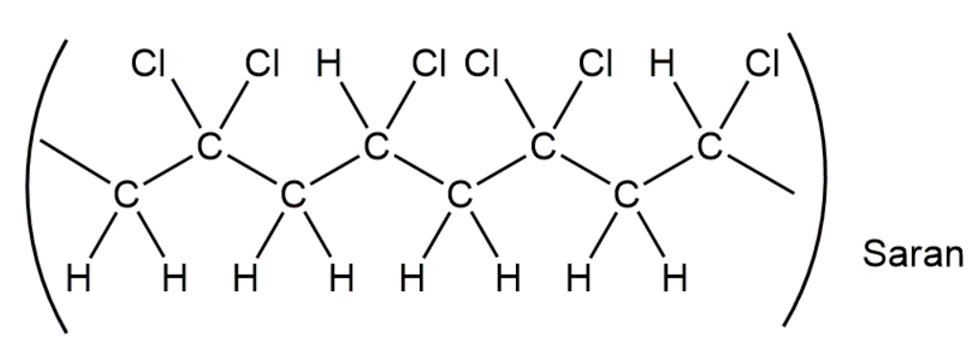
When a single alkene monomer, such as ethylene, is polymerized, the product is a homopolymer. If a mixture of two alkene monomers is polymerized, however, a copolymer often results. The following structure represents a segment of a copolymer called Saran. What two monomers were copolymerized to make Saran?
Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution:
What do you think about this solution?
We value your feedback to improve our textbook solutions.