Chapter 8: Q. 52a E (page 262)
Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution:
Short Answer
- But-1-ene
Learning Materials
Features
Discover
Chapter 8: Q. 52a E (page 262)
Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution:
All the tools & learning materials you need for study success - in one app.
Get started for free
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
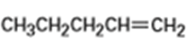
b)
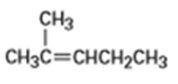
Compound A has the formula . On catalytic hydrogenation over palladium, it reacts with only 1 molar equivalent of . Compound A also undergoes reaction with ozone, followed by zinc treatment, to yield a symmetrical diketone, .
How many rings does A have?
Question:What product will result from hydroboration–oxidation of 1-methylcyclopentene with deuterated borane,? Show both the stereochemistry (spatial arrangement) and the regiochemistry (orientation) of the product.
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
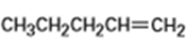
b)
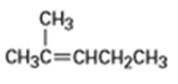
What alkenes might be used to prepare the following alcohols by hydroboration-oxidation?
What do you think about this solution?
We value your feedback to improve our textbook solutions.