Chapter 8: Q. 52b E (page 262)
Question:Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution
Short Answer
(b). 2 methyl pent-2-ene.
Learning Materials
Features
Discover
Chapter 8: Q. 52b E (page 262)
Question:Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution
(b). 2 methyl pent-2-ene.
All the tools & learning materials you need for study success - in one app.
Get started for free
Reaction of 2-methylpropene with CH3OH in the presence of H2SO4
catalyst yields methyl tert-butyl ether, CH3OC(CH3)3, by a mechanism
analogous to that of acid-catalyzed alkene hydration. Write the mechanism,
using curved arrows for each step.
How many alkene products, including E, Z isomers, might be obtained by dehydration of 3-methyl-3-hexanol with aqueous sulfuric acid?
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
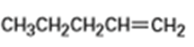
b)
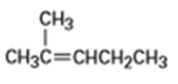
Compound A has the formula . On catalytic hydrogenation over palladium, it reacts with only 1 molar equivalent of . Compound A also reacts with ozone, followed by zinc treatment, to yield a symmetrical diketone, B.
Write the reactions.
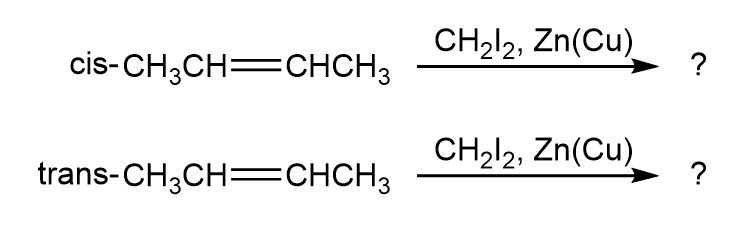
Question:The cis and trans isomers of 2-butene give different cyclopropane productsin the Simmons–Smith reaction. Show the structures of both, and
explain the difference.
What do you think about this solution?
We value your feedback to improve our textbook solutions.