Chapter 8: Q.52cE (page 262)
Question:Show the structures of alkenes that give the following products on oxidative cleavage with KMnO4 in acidic solution:
c.
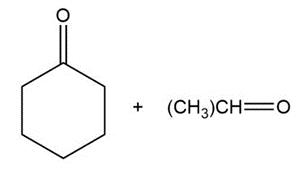
Short Answer
Answer
c.
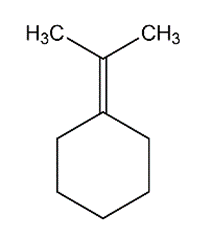
Learning Materials
Features
Discover
Chapter 8: Q.52cE (page 262)
Question:Show the structures of alkenes that give the following products on oxidative cleavage with KMnO4 in acidic solution:
c.

Answer
c.

All the tools & learning materials you need for study success - in one app.
Get started for free
We’ll see in the next chapter that alkynes undergo many of the same reactions that alkenes do. What product might you expect from each of the following reactions?
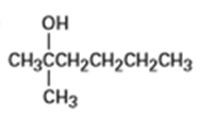
From what alkenes might the following alcohols have been prepared?
a)
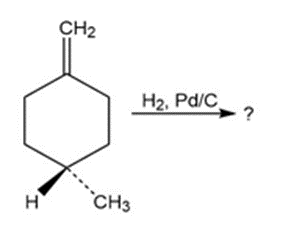
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
a)
An unknown hydrocarbon A with the formula reacts with 1 molar equivalent of over a palladium catalyst. Hydrocarbon A also reacts with to give diol B. When oxidized with in acidic solution, A gives two fragments. One fragment is propanoic acid, , and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction.
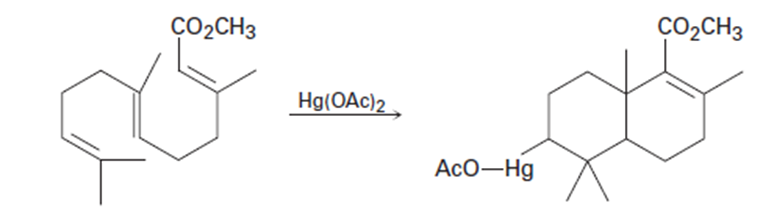
What do you think about this solution?
We value your feedback to improve our textbook solutions.