Chapter 8: Q. 70b E (page 262)
We’ll see in the next chapter that alkynes undergo many of the same reactions that alkenes do. What product might you expect from each of the following reactions?
Short Answer
(b).
Learning Materials
Features
Discover
Chapter 8: Q. 70b E (page 262)
We’ll see in the next chapter that alkynes undergo many of the same reactions that alkenes do. What product might you expect from each of the following reactions?
(b).
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Addition of HCl to 1,2-dimethylcyclohexene yields a mixture of two products. Show the stereochemistry of each, and explain why a mixture is formed.
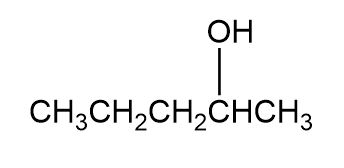
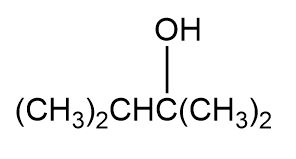
Question: Which of the following alcohols could notbe made selectively by hydroboration–oxidation of an alkene? Explain.
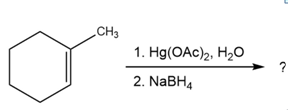
Question:Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate:
(d)
Question: Which of the following alcohols could notbe made selectively by hydroboration–oxidation of an alkene? Explain.
Compound A, C10H18O, undergoes reaction with dilute H2SO4at 50 °C to yield a mixture of two alkenes, C10H16. The major alkene product, B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Identify A and B, and write the reactions.
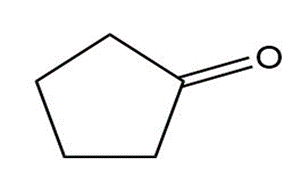
Cyclopentanone
What do you think about this solution?
We value your feedback to improve our textbook solutions.