Chapter 8: Q54b (page 262)
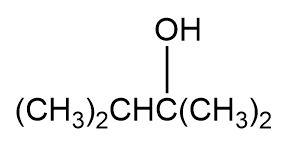
Question: Which of the following alcohols could notbe made selectively by hydroboration鈥搊xidation of an alkene? Explain.
Short Answer
Answer
Possible
Learning Materials
Features
Discover
Chapter 8: Q54b (page 262)
Question: Which of the following alcohols could notbe made selectively by hydroboration鈥搊xidation of an alkene? Explain.

Answer
Possible
All the tools & learning materials you need for study success - in one app.
Get started for free
Isolated from marine algae, prelaureatin is thought to be biosynthesized from laurediol by the following route. Propose a mechanism
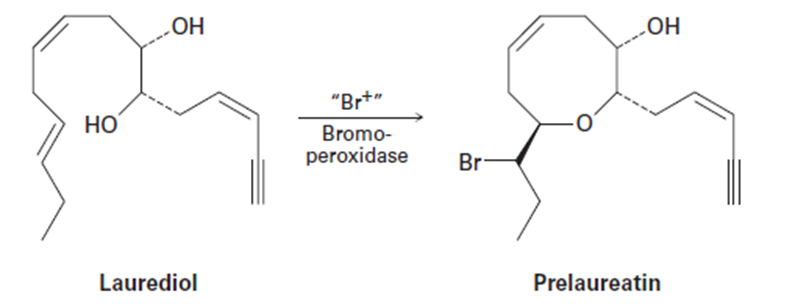
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction.
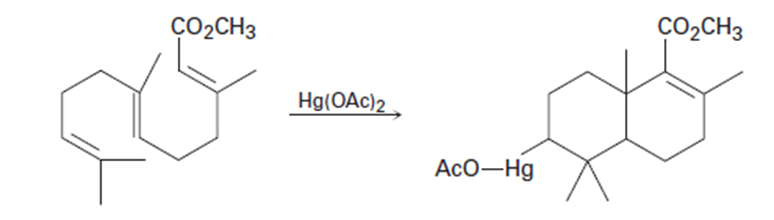
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
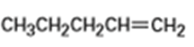
b)
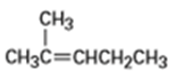
One problem with elimination reactions is that mixtures of products are often formed. For example, treatment of 2-bromo-2-methylbutane with KOH in ethanol yields a mixture of two alkene products. What are their likely structures?
Propose a curved-arrow mechanism to show how ozone (O3) reacts
with a carbon鈥揷arbon double bond to form a molozonide, the first intermediate
in ozonolysis.
What do you think about this solution?
We value your feedback to improve our textbook solutions.