Chapter 8: 8-7b (page 230)
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
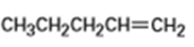
b)
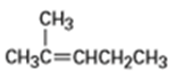
Short Answer
b) The product obtained from oxymercuration-demercuration of alkene is,
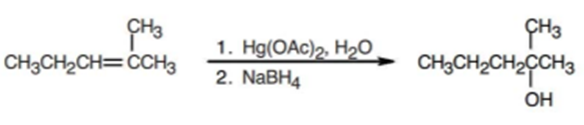
Learning Materials
Features
Discover
Chapter 8: 8-7b (page 230)
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
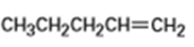
b)

b) The product obtained from oxymercuration-demercuration of alkene is,
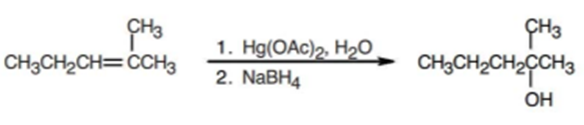
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What product would you expect to obtain from addition of to 1,2-dimethyl-cyclohexene? Show the stereochemistry of the product.
Compound A has the formula . On catalytic hydrogenation over palladium, it reacts with only 1 molar equivalent of . Compound A also undergoes a reaction with ozone, followed by zinc treatment, to yield a symmetrical diketone, B.
What are the structures of A and B?
Question:How would you carry out the following transformations? Tell what
reagents you would use in each case.
(a)
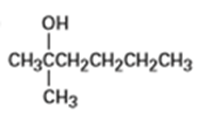
From what alkenes might the following alcohols have been prepared?
a)
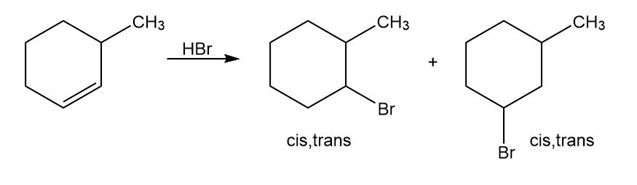
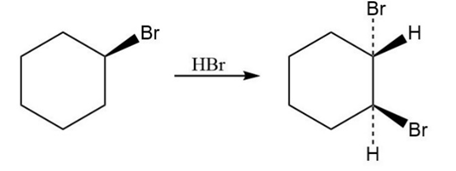
Reaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis- and trans-1-bromo-3-methylcyclohexane and cis- and trans-1-bromo-2-methylcyclohexane. The analogous reaction of HBr with 3-bromocyclohexene yields trans-1,2-dibromocyclohexane as the sole product. Draw structures of the possible intermediates, and then explain why only a single product is formed in this reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.