Chapter 8: Q50Ea (page 262)
Question:How would you carry out the following transformations? Tell what
reagents you would use in each case.
(a)
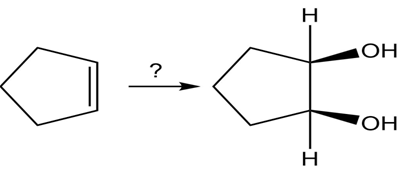
Short Answer
Answer
In this reaction, the reagent which we will use is :
Learning Materials
Features
Discover
Chapter 8: Q50Ea (page 262)
Question:How would you carry out the following transformations? Tell what
reagents you would use in each case.
(a)

Answer
In this reaction, the reagent which we will use is :

All the tools & learning materials you need for study success - in one app.
Get started for free
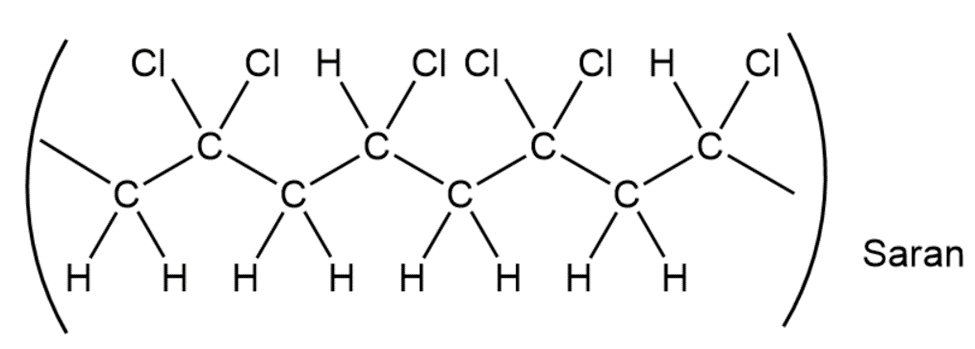
When a single alkene monomer, such as ethylene, is polymerized, the product is a homopolymer. If a mixture of two alkene monomers is polymerized, however, a copolymer often results. The following structure represents a segment of a copolymer called Saran. What two monomers were copolymerized to make Saran?
Compound A has the formula . On catalytic hydrogenation over palladium, it reacts with only 1 molar equivalent of . Compound A also reacts with ozone, followed by zinc treatment, to yield a symmetrical diketone, B.
Write the reactions.
What products are formed from hydration of 4-methyl cyclopentane? What can you say about the relative amounts of the products?
Draw the structure of a hydrocarbon that absorbs 2 molar equivalents of H2 on catalytic hydrogenation and gives only butanedial on ozonolysis.
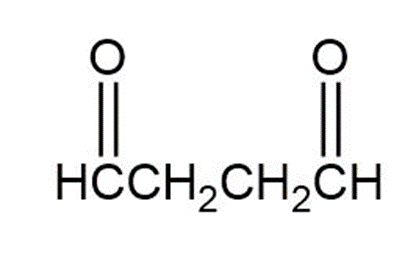
Butanedial
Hydroxylation of cis-2-butene with yields a different product than hydroxylation of trans-2-butene. Draw the structure, show the stereochemistry of each product, and explain the difference between them.
What do you think about this solution?
We value your feedback to improve our textbook solutions.