Chapter 8: Q54a (page 262)
Question: Which of the following alcohols could notbe made selectively by hydroboration鈥搊xidation of an alkene? Explain.
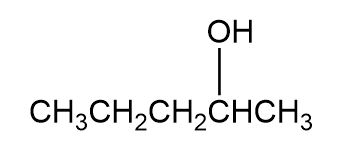
Short Answer
Answer
Not possible
Learning Materials
Features
Discover
Chapter 8: Q54a (page 262)
Question: Which of the following alcohols could notbe made selectively by hydroboration鈥搊xidation of an alkene? Explain.

Answer
Not possible
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What product would you expect to obtain from addition of to 1,2-dimethyl-cyclohexene? Show the stereochemistry of the product.
The following alkene undergoes hydroboration鈥搊xidation to yield a single product rather than a mixture. Explain the result, and draw the product showing its stereochemistry.
What product would you expect from the reaction of cyclopentene with NBS and water? Show the stereochemistry.
Dichlorocarbene can be generated by heating sodium trichloroacetate.
Propose a mechanism for the reaction, and use curved arrows to indicate
the movement of electrons in each step. What relationship does
your mechanism bear to the base-induced elimination of HCl from
chloroform?

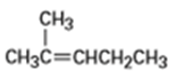
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
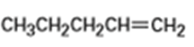
b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.