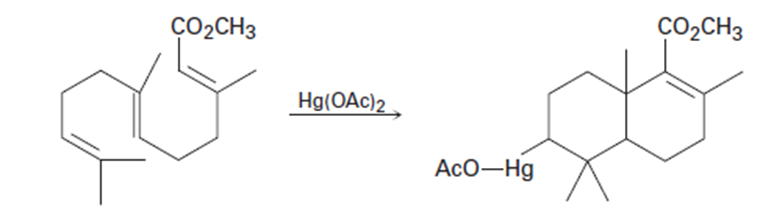
Chapter 8: Q44Ed (page 262)
Question:Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate:
(d)
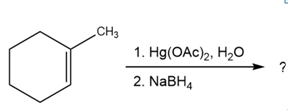
Short Answer
Answer
The product of the reaction is:
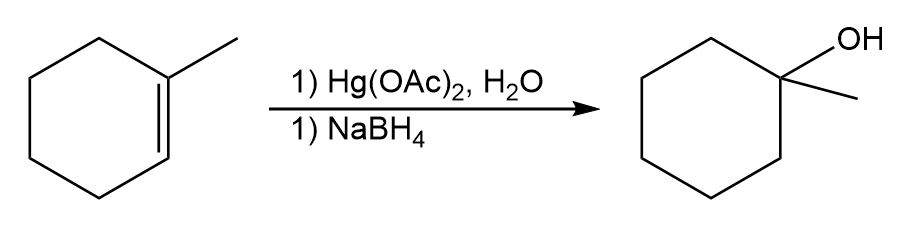
Learning Materials
Features
Discover
Chapter 8: Q44Ed (page 262)
Question:Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate:
(d)

Answer
The product of the reaction is:
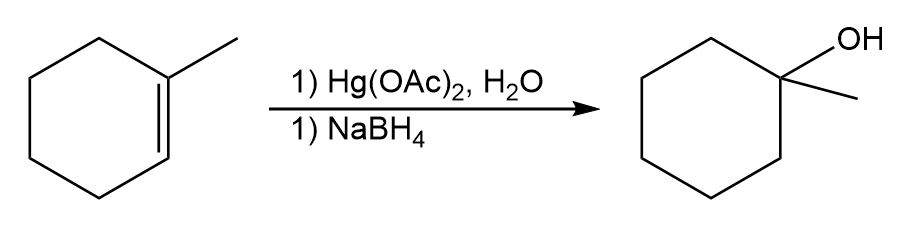
All the tools & learning materials you need for study success - in one app.
Get started for free
Show the structures of alkenes that give the following products on oxidative cleavage within acidic solution:
One problem with elimination reactions is that mixtures of products are often formed. For example, treatment of 2-bromo-2-methylbutane with KOH in ethanol yields a mixture of two alkene products. What are their likely structures?
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction.
The following alkene undergoes hydroboration–oxidation to yield a single product rather than a mixture. Explain the result, and draw the product showing its stereochemistry.
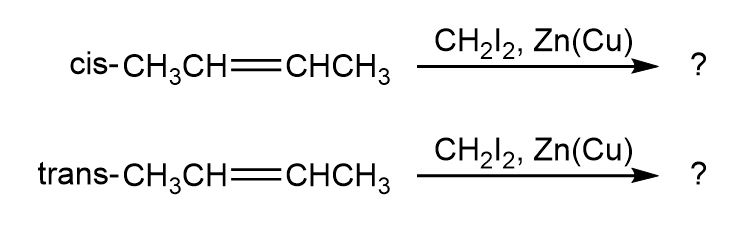
Question:The cis and trans isomers of 2-butene give different cyclopropane productsin the Simmons–Smith reaction. Show the structures of both, and
explain the difference.
What do you think about this solution?
We value your feedback to improve our textbook solutions.