Chapter 8: Q52cE. (page 262)
Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution.
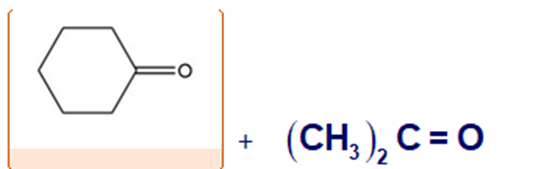
Short Answer
Learning Materials
Features
Discover
Chapter 8: Q52cE. (page 262)
Show the structures of alkenes that give the following products on oxidative cleavage with in acidic solution.
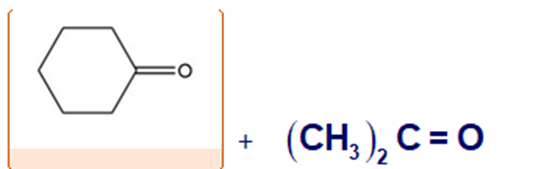

All the tools & learning materials you need for study success - in one app.
Get started for free
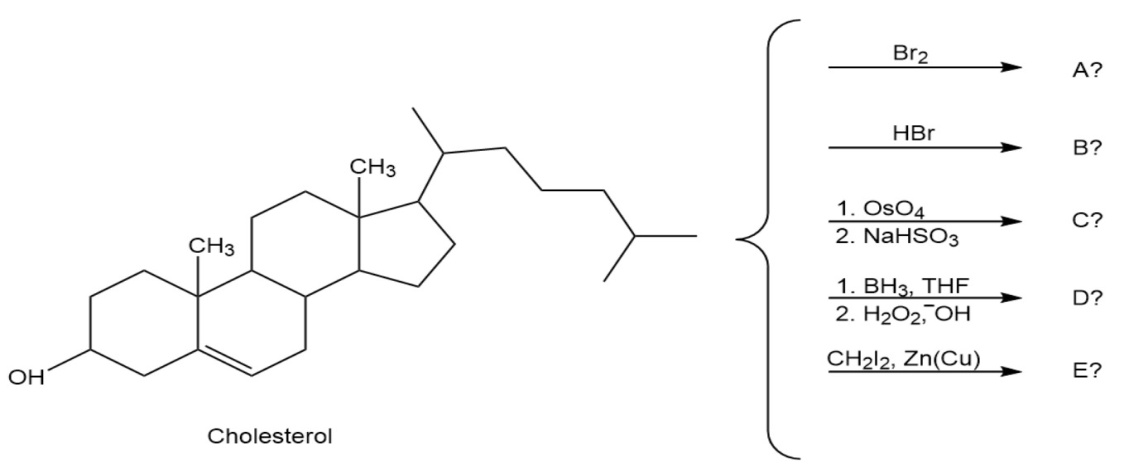
Question:Predict the products of the following reactions. Don’t worry about thesize of the molecule; concentrate on the functional groups.
What alkenes might be used to prepare the following alcohols by hydroboration-oxidation?
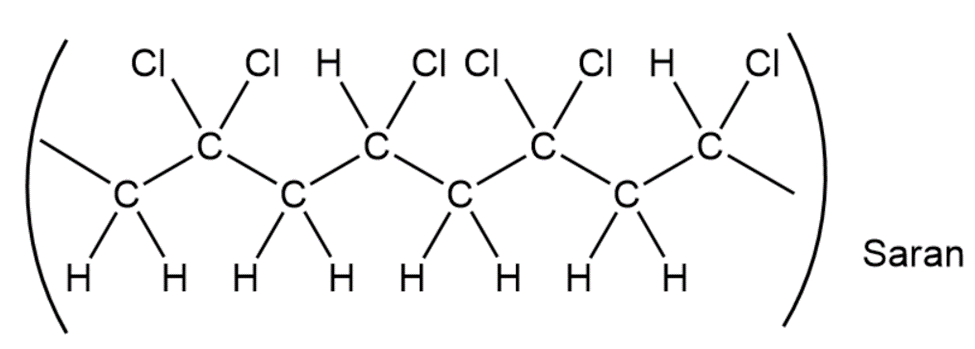
When a single alkene monomer, such as ethylene, is polymerized, the product is a homopolymer. If a mixture of two alkene monomers is polymerized, however, a copolymer often results. The following structure represents a segment of a copolymer called Saran. What two monomers were copolymerized to make Saran?
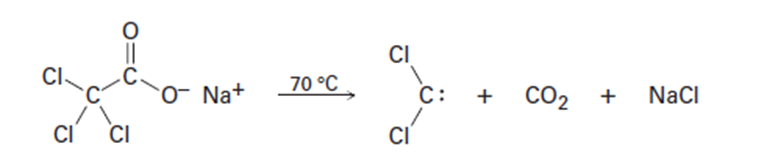
Dichlorocarbene can be generated by heating sodium trichloroacetate.
Propose a mechanism for the reaction, and use curved arrows to indicate
the movement of electrons in each step. What relationship does
your mechanism bear to the base-induced elimination of HCl from
chloroform?
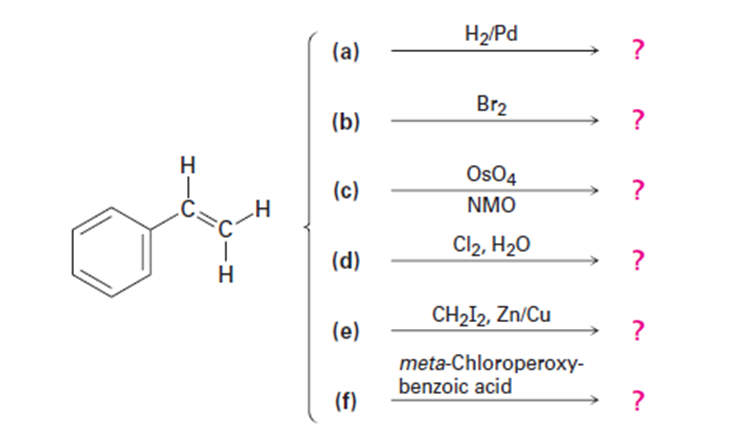
Predict the products of the following reactions (the aromatic ring is
unreactive in all cases). Indicate regiochemistry when relevant.
What do you think about this solution?
We value your feedback to improve our textbook solutions.