Chapter 8: Q48E (page 262)
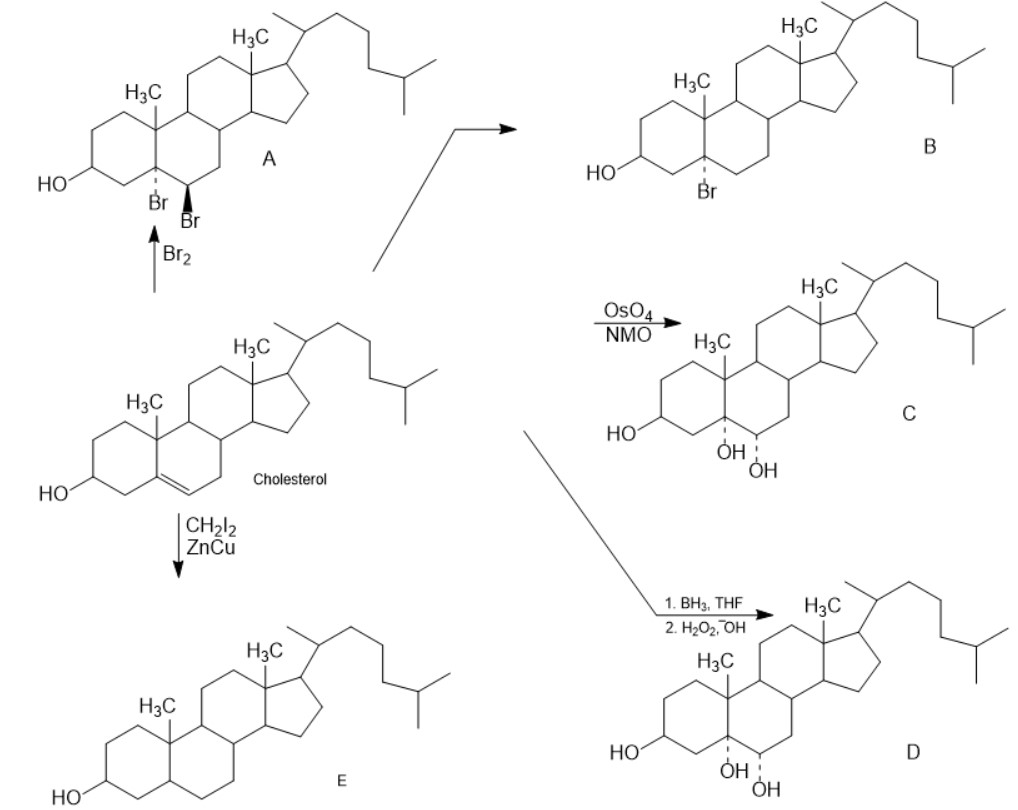
Question:Predict the products of the following reactions. Don’t worry about thesize of the molecule; concentrate on the functional groups.
Short Answer
Answer
Products of the reactions are:
Learning Materials
Features
Discover
Chapter 8: Q48E (page 262)
Question:Predict the products of the following reactions. Don’t worry about thesize of the molecule; concentrate on the functional groups.

Answer
Products of the reactions are:

All the tools & learning materials you need for study success - in one app.
Get started for free
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
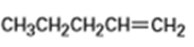
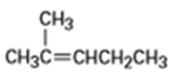
b)
Hydroxylation of cis-2-butene with yields a different product than hydroxylation of trans-2-butene. Draw the structure, show the stereochemistry of each product, and explain the difference between them.
Question: Addition of HCl to 1,2-dimethylcyclohexene yields a mixture of two products. Show the stereochemistry of each, and explain why a mixture is formed.
Question: Which of the following alcohols could notbe made selectively by hydroboration–oxidation of an alkene? Explain.
Compound A has the formula . It reacts rapidly with to give and a carboxylic acid, B , but reacts with only 1 molar equivalent of on catalytic hydrogenation over a palladium catalyst. On hydrogenation under conditions that reduce aromatic rings, 4 equivalents of are taken up and hydrocarbon C is produced. What are the structures of A, B, and C? Write the reactions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.