Chapter 8: Q10P (page 234)
What alkenes might be used to prepare the following alcohols by hydroboration-oxidation?
Learning Materials
Features
Discover
Chapter 8: Q10P (page 234)
What alkenes might be used to prepare the following alcohols by hydroboration-oxidation?
All the tools & learning materials you need for study success - in one app.
Get started for free
Simmons–Smith reaction of cyclohexene with diiodomethane gives asingle cyclopropane product, but the analogous reaction of cyclohexene with 1,1-diiodoethane gives (in low yield) a mixture of two isomeric methylcyclopropane products. What are the two products, and how do they differ?
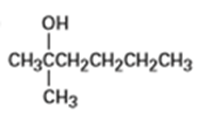
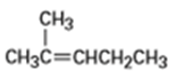
From what alkenes might the following alcohols have been prepared?
a)
Question:Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate:
(d)
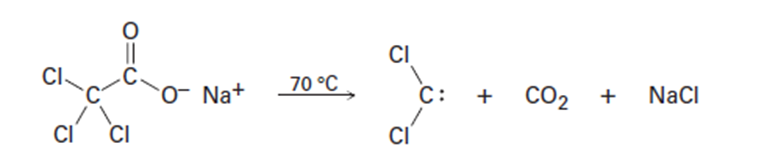
Dichlorocarbene can be generated by heating sodium trichloroacetate.
Propose a mechanism for the reaction, and use curved arrows to indicate
the movement of electrons in each step. What relationship does
your mechanism bear to the base-induced elimination of HCl from
chloroform?
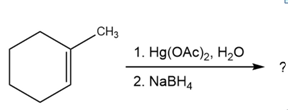
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
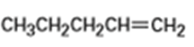
b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.