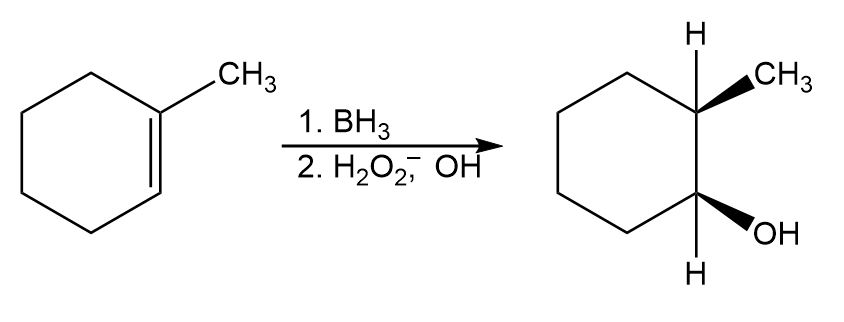
Chapter 8: Q11P (page 234)
The following cycloalkene gives a mixture of two alcohols on hydroboration followed by oxidation. Draw the structure of both, and explain the result.
Learning Materials
Features
Discover
Chapter 8: Q11P (page 234)
The following cycloalkene gives a mixture of two alcohols on hydroboration followed by oxidation. Draw the structure of both, and explain the result.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What product would you expect to obtain from addition of to 1,2-dimethyl-cyclohexene? Show the stereochemistry of the product.
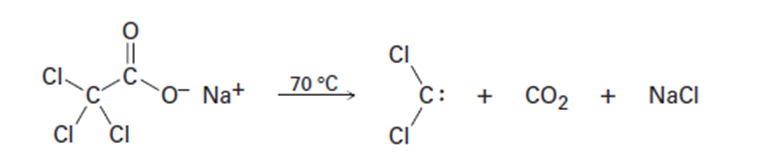
Question: In planning the synthesis of one compound from another, it’s just as important to know what not to do as to know what to do. The following reactions all have serious drawbacks to them. Explain the potential problems of each.
Dichlorocarbene can be generated by heating sodium trichloroacetate.
Propose a mechanism for the reaction, and use curved arrows to indicate
the movement of electrons in each step. What relationship does
your mechanism bear to the base-induced elimination of HCl from
chloroform?
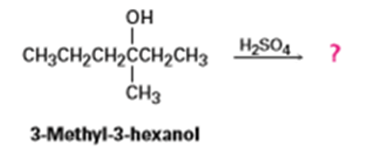
Question: How many alkene products, including E, Z isomers, might be obtained by dehydration of 3-methyl-3-hexanol with aqueous sulfuric acid?
What alkenes might be used to prepare the following alcohols by hydroboration-oxidation?
What do you think about this solution?
We value your feedback to improve our textbook solutions.