Chapter 8: Q8-2P. (page 222)
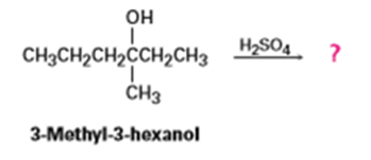
Question: How many alkene products, including E, Z isomers, might be obtained by dehydration of 3-methyl-3-hexanol with aqueous sulfuric acid?
Short Answer
Answer
The five alkene products formed are,
Learning Materials
Features
Discover
Chapter 8: Q8-2P. (page 222)
Question: How many alkene products, including E, Z isomers, might be obtained by dehydration of 3-methyl-3-hexanol with aqueous sulfuric acid?

Answer
The five alkene products formed are,

All the tools & learning materials you need for study success - in one app.
Get started for free
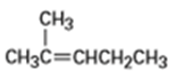
What products would you expect from oxymercuration-demercuration of the following alkenes?
a)
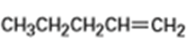
b)
Compound A, C10H18O, undergoes reaction with dilute H2SO4at 50 °C to yield a mixture of two alkenes, C10H16. The major alkene product, B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Identify A and B, and write the reactions.
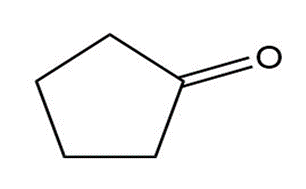
Cyclopentanone
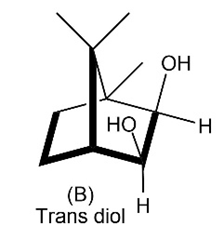
Evidence that cleavage of 1,2-diols by occurs through a five membered cyclic periodate intermediate is based on kinetic data—the measurement of reaction rates. When diols A and B were prepared and the rates of their reaction withwere measured, it was found that diol A cleaved approximately 1 million times faster than diol B. Make molecular models of A and B and of potential cyclic periodate intermediates, and then explain the kinetic results

An unknown hydrocarbon A with the formula reacts with 1 molar equivalent of over a palladium catalyst. Hydrocarbon A also reacts with to give diol B. When oxidized with in acidic solution, A gives two fragments. One fragment is propanoic acid, , and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.
Hydroxylation of cis-2-butene with yields a different product than hydroxylation of trans-2-butene. Draw the structure, show the stereochemistry of each product, and explain the difference between them.
What do you think about this solution?
We value your feedback to improve our textbook solutions.