Chapter 8: 8-34b (page 262)
Iodine azide,adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as1-butene isused, only one product results:
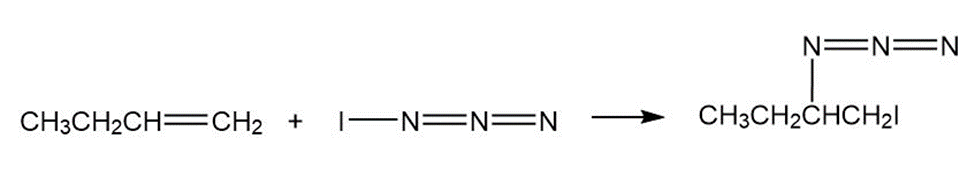
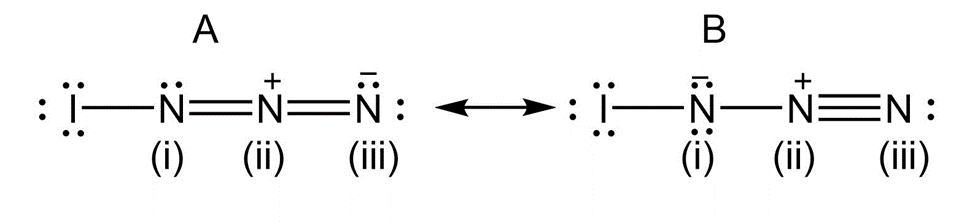
b) Calculate formal charges for the atoms in both resonance structuresyou drew for,in part (a).
Short Answer
Formal charge for both the resonance structures are:
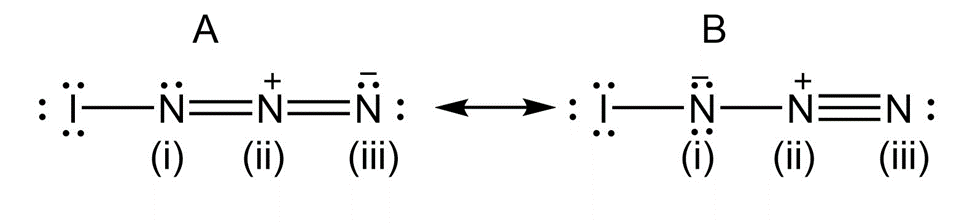
Formal Charge
I N(i) N(ii) N(iii)
A) 0 0 +1 -1
B) 0 -1 +1 0