Chapter 8: 8-32c (page 262)
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
c)
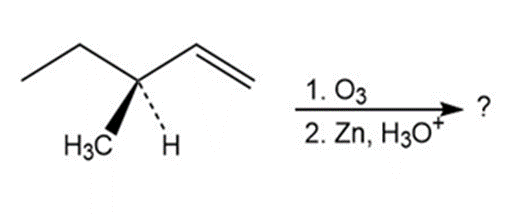
Short Answer
The product mixture of this reaction will rotate plane polarized light.
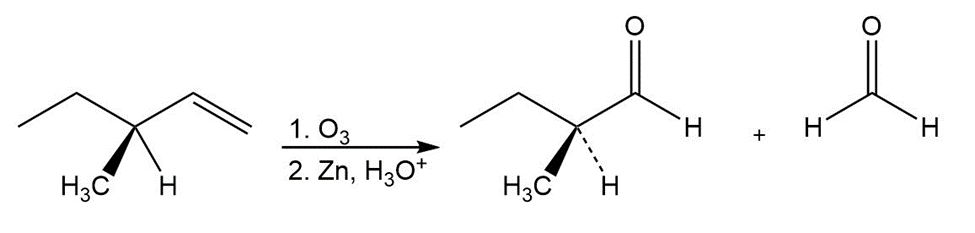
Learning Materials
Features
Discover
Chapter 8: 8-32c (page 262)
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
c)

The product mixture of this reaction will rotate plane polarized light.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: In planning the synthesis of one compound from another, it’s just as important to know what not to do as to know what to do. The following reactions all have serious drawbacks to them. Explain the potential problems of each.
When an unsymmetrical alkene such as propene is treated with N-bromosuccinimide in aqueous dimethyl sulfoxide, the major product has the bromine atom bonded to the less highly substituted carbon atom. Is this Markovnikov or non-Markovnikov orientation? Explain
Question:How would you carry out the following transformations? Tell what
reagents you would use in each case.
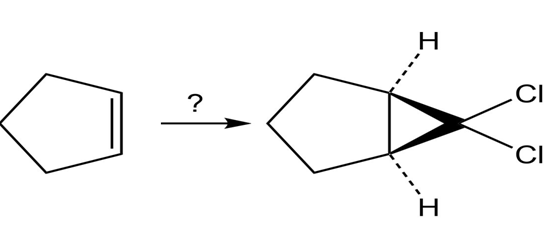
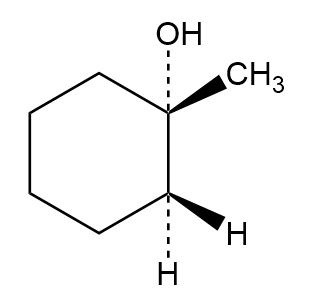
Question: Which of the following alcohols could notbe made selectively by hydroboration–oxidation of an alkene? Explain.
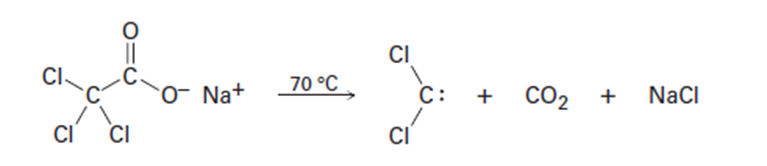
Dichlorocarbene can be generated by heating sodium trichloroacetate.
Propose a mechanism for the reaction, and use curved arrows to indicate
the movement of electrons in each step. What relationship does
your mechanism bear to the base-induced elimination of HCl from
chloroform?
What do you think about this solution?
We value your feedback to improve our textbook solutions.