Chapter 8: 8-32b (page 262)
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
b)
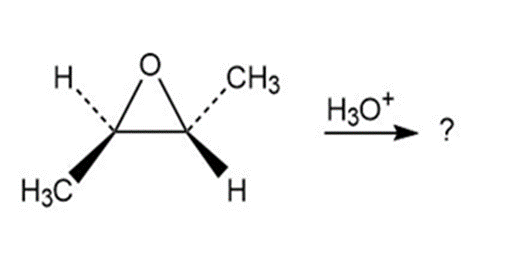
Short Answer
The product of this reaction will not rotate plane polarized light.
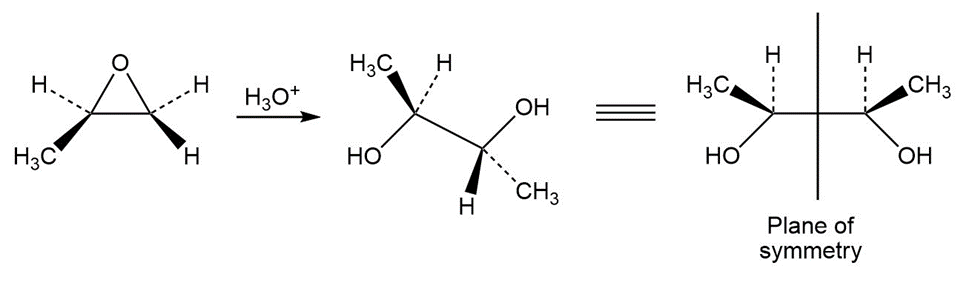
Learning Materials
Features
Discover
Chapter 8: 8-32b (page 262)
Which of the reactions below would result in a product mixture thatwould rotate plane-polarized light?
b)

The product of this reaction will not rotate plane polarized light.

All the tools & learning materials you need for study success - in one app.
Get started for free
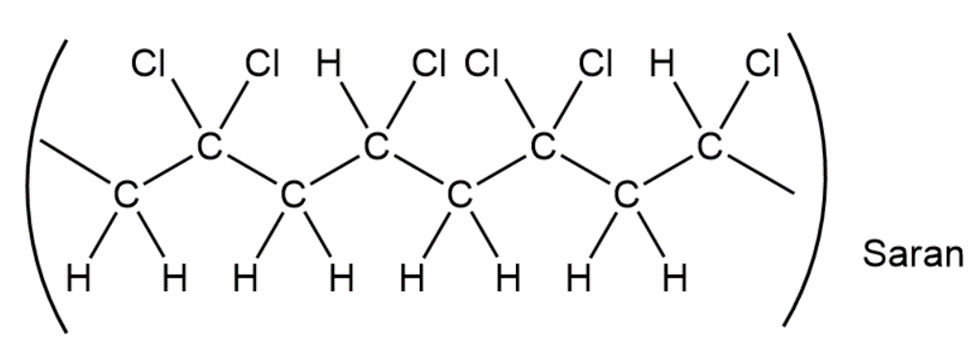
When a single alkene monomer, such as ethylene, is polymerized, the product is a homopolymer. If a mixture of two alkene monomers is polymerized, however, a copolymer often results. The following structure represents a segment of a copolymer called Saran. What two monomers were copolymerized to make Saran?
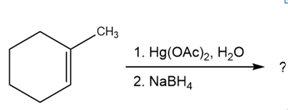
Question:Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate:
(d)
Compound A has the formula . On catalytic hydrogenation over palladium, it reacts with only 1 molar equivalent of . Compound A also undergoes reaction with ozone, followed by zinc treatment, to yield a symmetrical diketone, .
How many rings does A have?
Question: Which of the following alcohols could notbe made selectively by hydroboration–oxidation of an alkene? Explain.
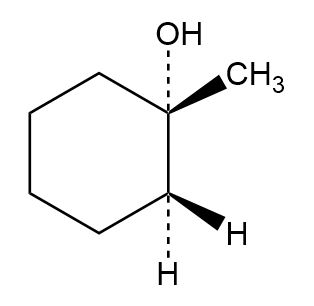
Question: Addition of HCl to 1,2-dimethylcyclohexene yields a mixture of two products. Show the stereochemistry of each, and explain why a mixture is formed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.