Chapter 1: Q1-1-42E-a (page 27)
Convert the following structures into skeletal drawings
(a)
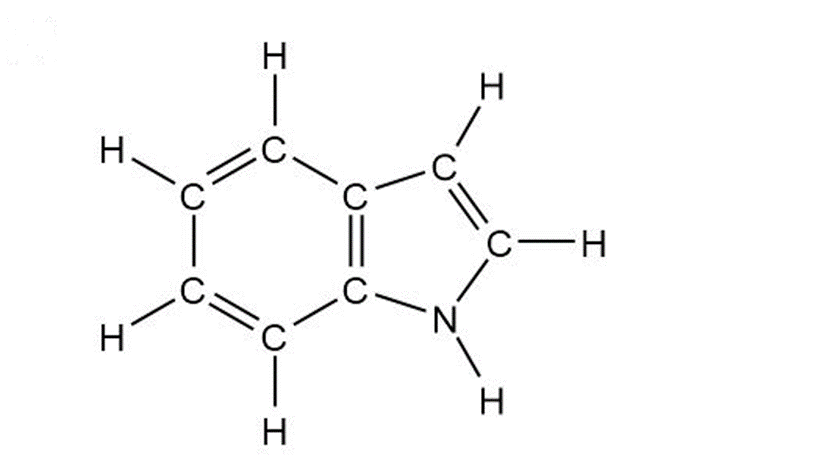
Indole
Short Answer
(a)
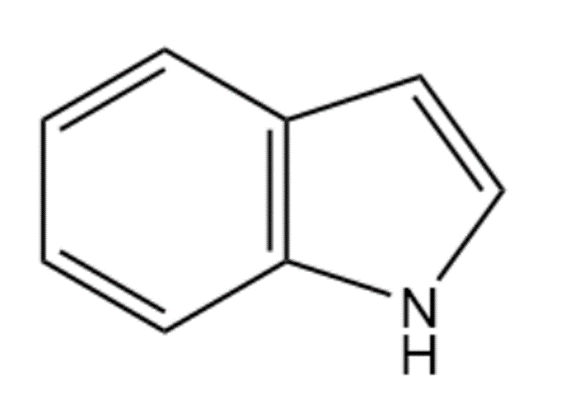
Skeletal structure of Indole
Learning Materials
Features
Discover
Chapter 1: Q1-1-42E-a (page 27)
Convert the following structures into skeletal drawings
(a)

Indole
(a)

Skeletal structure of Indole
All the tools & learning materials you need for study success - in one app.
Get started for free
There are three different substances that contain a carbon–carbon double bond and have the formula C4H8. Draw them, and tell how they differ.
Convert the following molecular model of hexane, a component of gasoline, into a line-bond structure (gray 5 C, ivory 5 H).
 HEXANE
HEXANE
What are likely formulas for the following molecules?
(a) NH?OH (b) AlCl?(c) CF2Cl(d)CHO?
(b)
Allene, is somewhat unusual in that it has two adjacent double bonds. Draw a picture showing the orbitals involved in the s and p bonds of allene. Is the central carbon atomsp2orsp-hybridized? What about the hybridization of the terminal carbons? What shape do you predict for allene?
Draw a line-bond structure for propane, . Predict the value of each bond angle, and indicate the overall shape of the molecule.
What do you think about this solution?
We value your feedback to improve our textbook solutions.