Chapter 1: Q1-1-35E (page 27)
What is the hybridization of each carbon atom in acetonitrile?
Short Answer
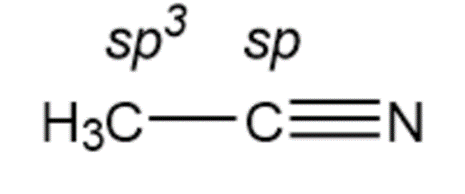
Hybridization of carbon in acetonitrile
Learning Materials
Features
Discover
Chapter 1: Q1-1-35E (page 27)
What is the hybridization of each carbon atom in acetonitrile?
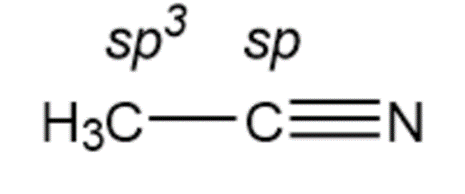
Hybridization of carbon in acetonitrile
All the tools & learning materials you need for study success - in one app.
Get started for free
Give the ground-state electron configuration for each of the following elements:
(a)Potassium (b) Arsenic (c) Aluminum (d) Germanium
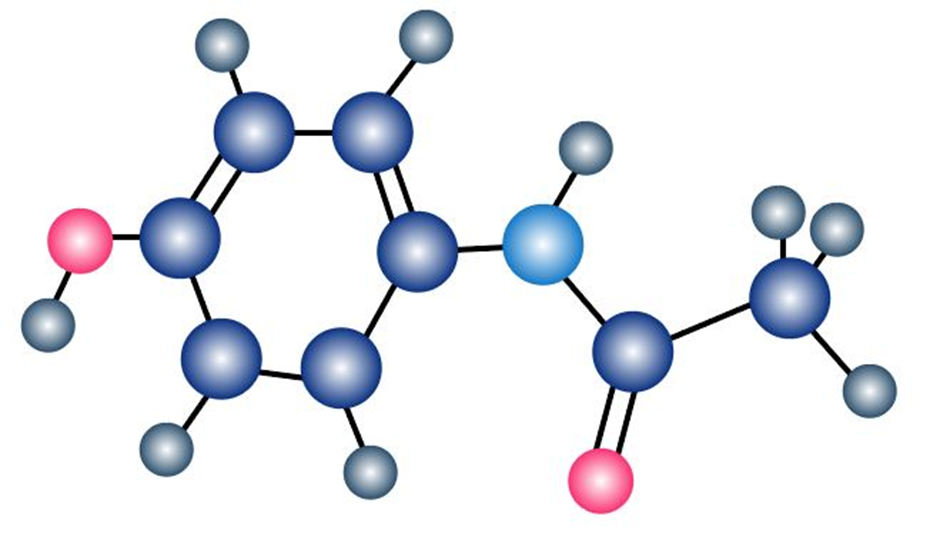
The following model is a representation of acetaminophen, a pain reliever sold in drugstores under a variety of names, including Tylenol. Identify the hybridization of each carbon atom in acetaminophen, and tell which atoms have lone pairs of electrons (gray 5 C, red 5 O, blue 5 N, ivory 5 H).
Question: Convert each of the following molecular models into a skeletal structure, and give the formula of each. Only the connections between atoms
are shown; multiple bonds are not indicated (gray 5 C, red 5 O, blue 5 N, ivory 5 H).
(a)
(b)
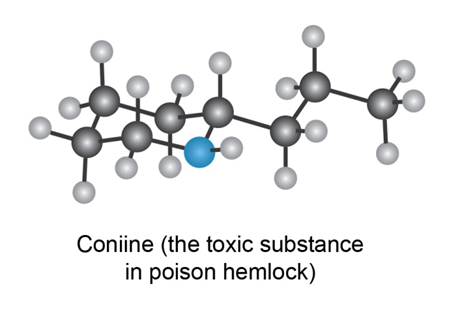
Propose skeletal structures for compounds that satisfy the following molecular formulas. There is more than one possibility in each case.
(a)
(b)
(c)
(d)
Why can’t an organic molecule have the formula (C2H7)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.