Chapter 1: Q1-1-7P (page 10)
Why can’t an organic molecule have the formula (C2H7)?
Short Answer
An organic molecule cannot have the formula because that would require a C atom to have more than 4 bonds or an H atom to have more than 1 bond.
Learning Materials
Features
Discover
Chapter 1: Q1-1-7P (page 10)
Why can’t an organic molecule have the formula (C2H7)?
An organic molecule cannot have the formula because that would require a C atom to have more than 4 bonds or an H atom to have more than 1 bond.
All the tools & learning materials you need for study success - in one app.
Get started for free
How many valence electrons does each of the following dietary trace elements have?
(a) Zinc (b) Iodine (c) Silicon (d) Iron
Oxaloacetic acid, an important intermediate in food metabolism, has the formulaand contains threebonds and twobonds. Propose two possible structures.
Pyridoxal phosphate, a close relative of vitaminB6, is involved in a large number of metabolic reactions. Tell the hybridization, and predict the bond angles for each nonterminal atom?
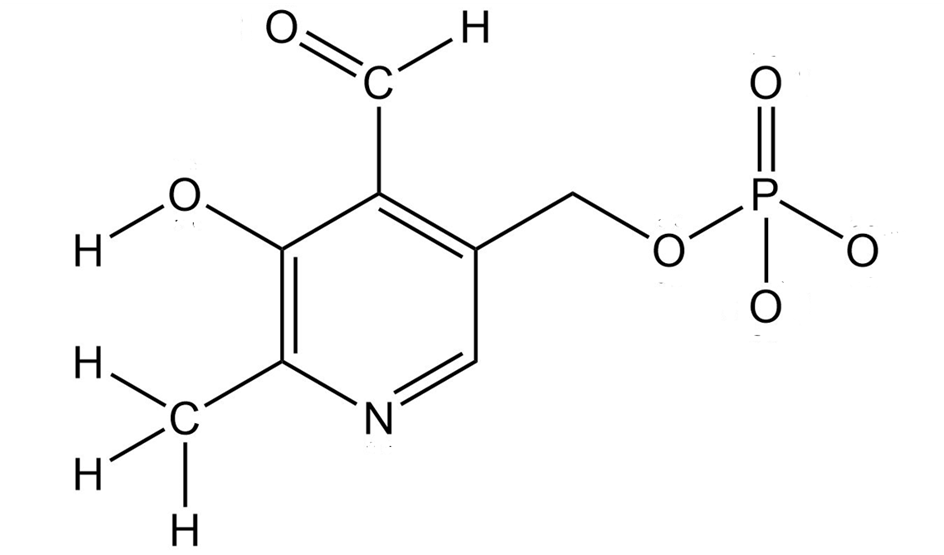
Pyridoxal phosphate
Give the ground-state electron configuration for each of the following elements:
(a) Oxygen (b) Nitrogen (c) Sulfur
Potassium methoxide,,KOCH3 contains both covalent and ionic bonds. Which do you think is which?
What do you think about this solution?
We value your feedback to improve our textbook solutions.