Chapter 16: Q16-18P (page 511)
What aromatic products would you obtain from the oxidation of the following substances?
a.
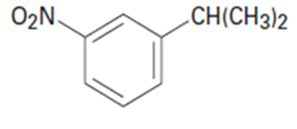
b.
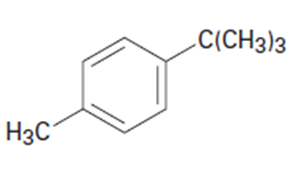
Short Answer
a.
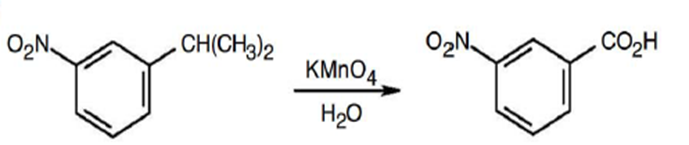
b.
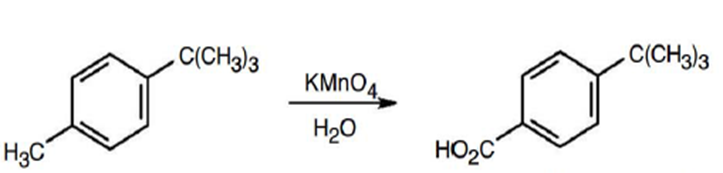
Learning Materials
Features
Discover
Chapter 16: Q16-18P (page 511)
What aromatic products would you obtain from the oxidation of the following substances?
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
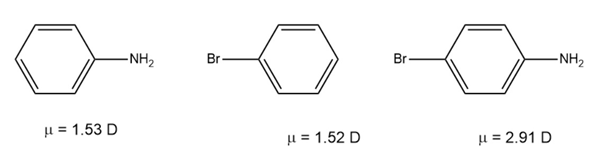
Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline andbromobenzene.
Question: Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.
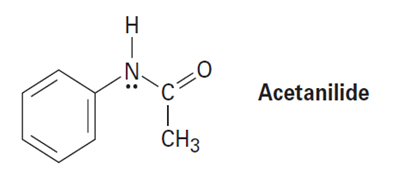
Acetanilide is less reactive than aniline toward electrophilic substitution. Explain
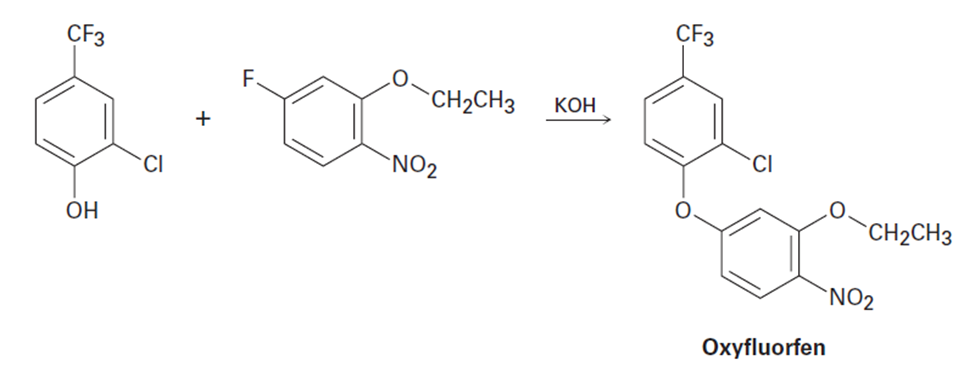
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose a mechanism.
Would you expect the Friedel–Crafts reaction of benzene with (R)-2-chlorobutane to yield optically active or racemic product? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.