Chapter 16: Q73E (page 524)
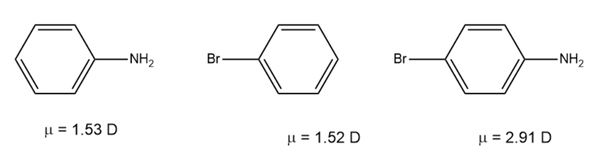
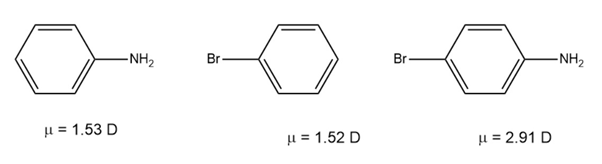
Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline andbromobenzene.
Short Answer
Directions of the dipole moment as:
Learning Materials
Features
Discover
Chapter 16: Q73E (page 524)
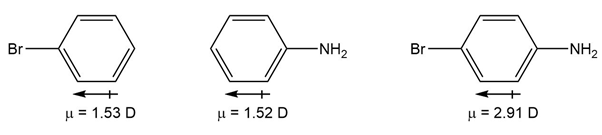
Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline andbromobenzene.

Directions of the dipole moment as:

All the tools & learning materials you need for study success - in one app.
Get started for free
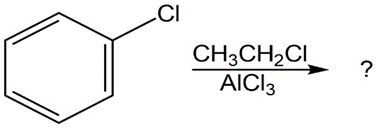
Question:Predict the major product(s) of the following reactions:
(a)
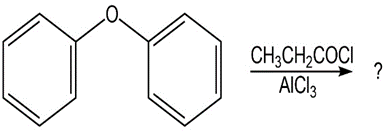
(b)
(c)
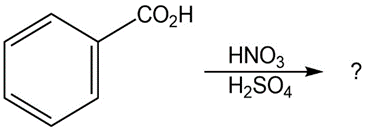
(d)
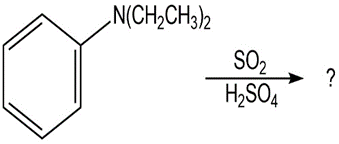
Question: Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline and bromobenzene.
Monobromination of toluene gives a mixture of three bromotoluene products. Draw and name them.
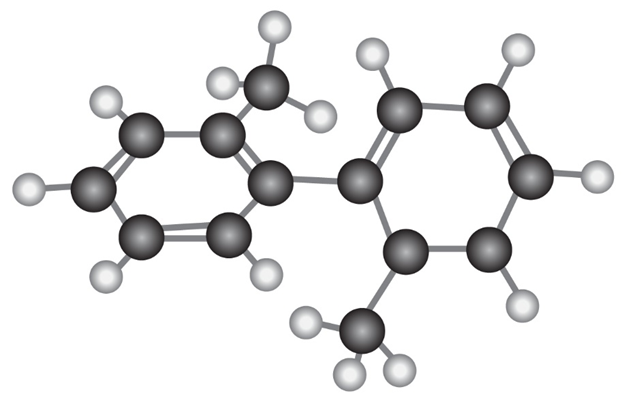
The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy conformation of the molecule. Why are the two benzene rings tilted at a 63° angle to each other rather than being in the same plane so that their p orbitals overlap? Why doesn’t complete rotation around the single bond joining the two rings occur?
At what position, and on what ring, would you expect the followingsubstances to undergo electrophilic substitution?
(a)
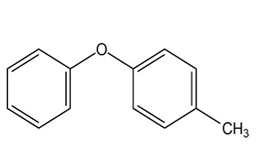
(b)
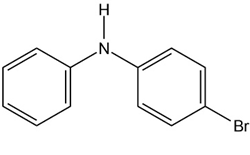
(c)
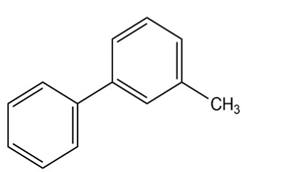
(d)
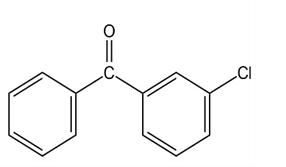
What do you think about this solution?
We value your feedback to improve our textbook solutions.