Chapter 16: Q16-1P (page 482)
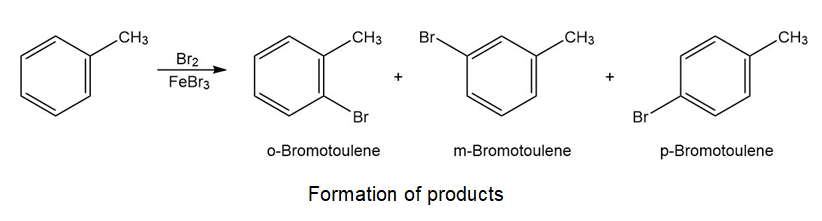
Monobromination of toluene gives a mixture of three bromotoluene products. Draw and name them.
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q16-1P (page 482)
Monobromination of toluene gives a mixture of three bromotoluene products. Draw and name them.

All the tools & learning materials you need for study success - in one app.
Get started for free
The nitroso group, - NO, is one of the few non-halogens that is an ortho- and para-directing deactivator. Explain this behaviour by drawing resonance structures of the carbocation intermediates in ortho, meta, and para electrophilic reaction on nitroso benzene,.
Rank the compounds in each group according to their reactivity toward electrophilic substitution.
At what position, and on what ring, would you expect bromination of Benzanilide to occur? Explain by drawing resonance structures of theintermediates.
 Benzanilide
Benzanilide
Refer to Table 6-3 on page 170 for a quantitative idea of the stability of a benzyl radical. How much more stable (in kJ/mol) is the benzyl radical than a primary alkyl radical? How does a benzyl radical compare in stability to an allylradical?
Starting with benzene as your only source of aromatic compounds,
how would you synthesize the following substances? Assume that you
can separate ortho and para isomers if necessary.
(a)p-Chloroacetophenone (b)m-Bromonitrobenzene
(c)o-Bromobenzenesulfonic acid (d)m-Chlorobenzenesulfonic acid
What do you think about this solution?
We value your feedback to improve our textbook solutions.