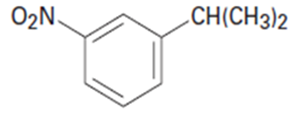
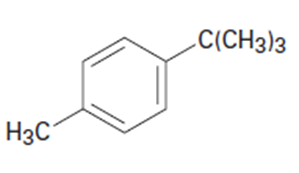
Chapter 16: Q56E (page 524)
Starting with benzene as your only source of aromatic compounds,
how would you synthesize the following substances? Assume that you
can separate ortho and para isomers if necessary.
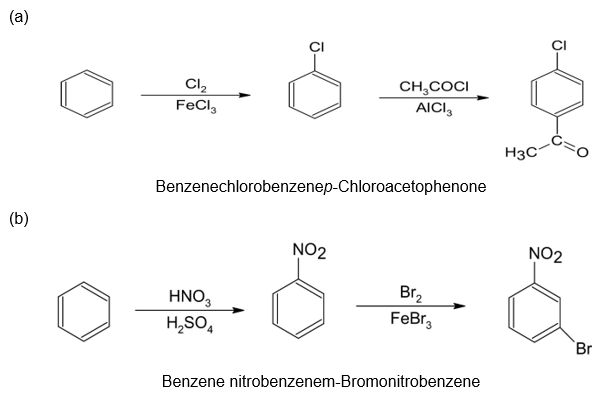
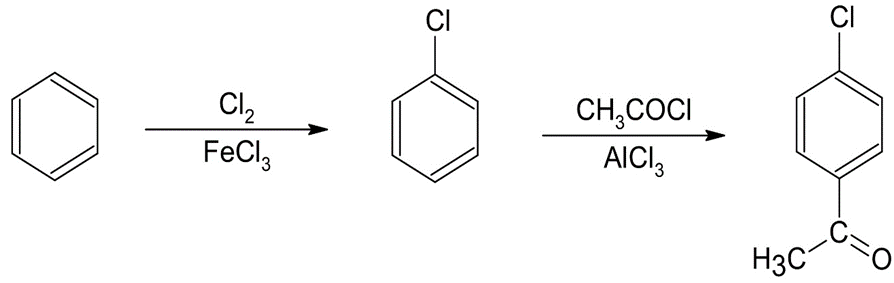
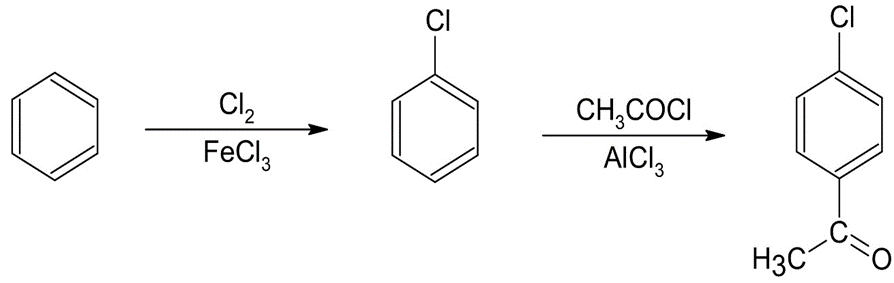
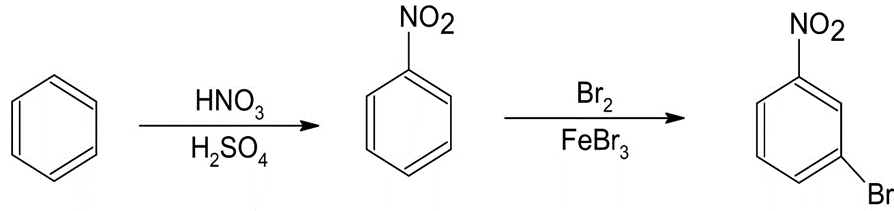
(a)p-Chloroacetophenone (b)m-Bromonitrobenzene
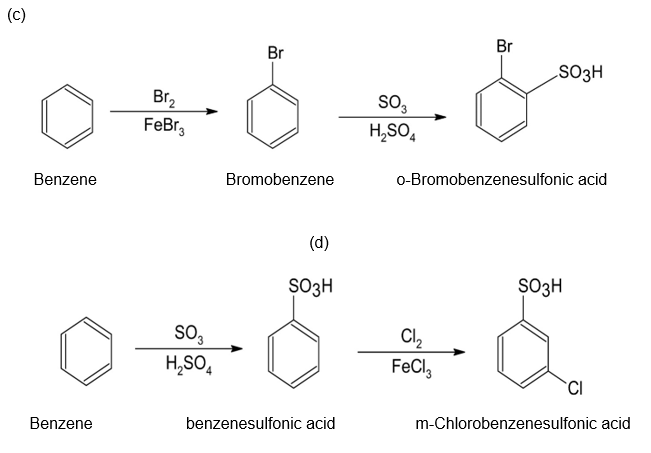
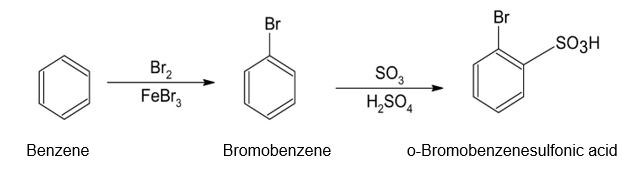
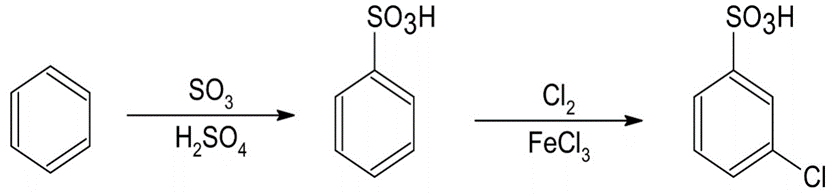
(c)o-Bromobenzenesulfonic acid (d)m-Chlorobenzenesulfonic acid
Short Answer
Answer
The synthesis of benzene gives the following product: