Chapter 16: Q6 P (page 493)
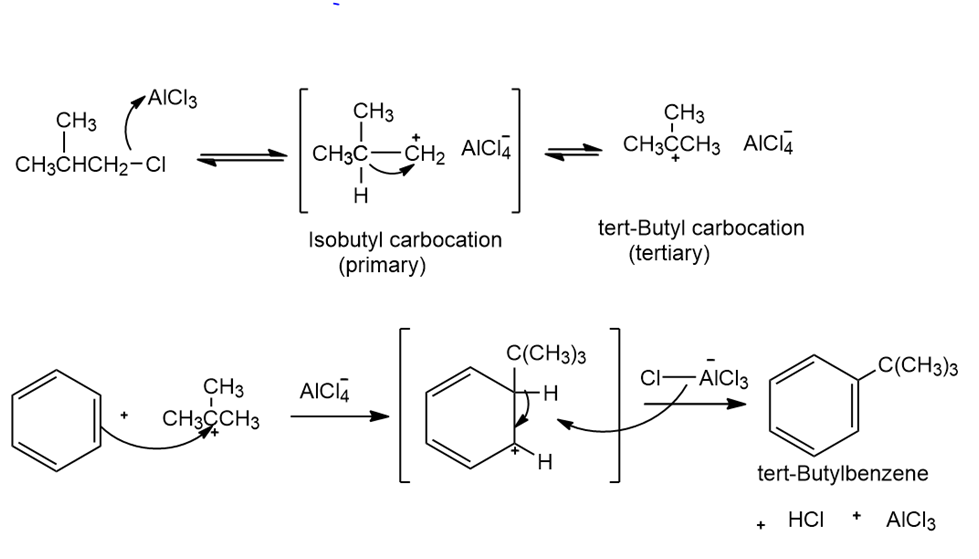
What is the major monosubstitution product from the Friedel–Crafts reaction of benzene with 1-chloro-2-methylpropane in the presence of ?
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q6 P (page 493)
What is the major monosubstitution product from the Friedel–Crafts reaction of benzene with 1-chloro-2-methylpropane in the presence of ?

All the tools & learning materials you need for study success - in one app.
Get started for free
What product(s) would you expect to obtain from the following
reactions?
(a)
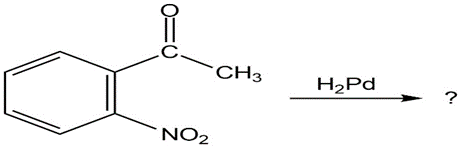
(b)
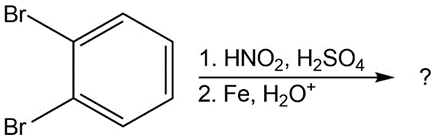
(c)
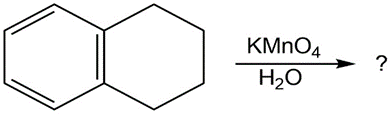
(d)
How many products might be formed on chlorination of o-xylene (o-dimethylbenzene), m-xylene, and p-xylene?
p-Bromotoluene reacts with potassium amide to give a mixture of mand p-methylaniline. Explain.
Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.
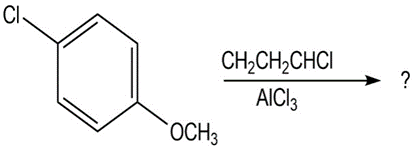
Question:Predict the major product(s) of the following reactions:
(a)
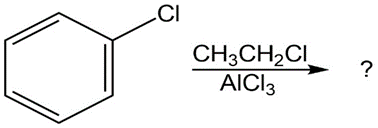
(b)
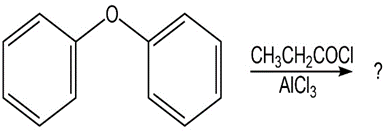
(c)
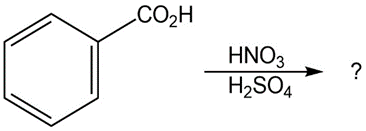
(d)
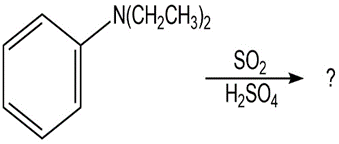
What do you think about this solution?
We value your feedback to improve our textbook solutions.