Chapter 16: Q 3P (page 486)
How many products might be formed on chlorination of o-xylene (o-dimethylbenzene), m-xylene, and p-xylene?
Short Answer
 Chlorination of o-xylene
Chlorination of o-xylene
 Chlorination of meta-xylene
Chlorination of meta-xylene
 Chlorination of para-xylene
Chlorination of para-xylene
Learning Materials
Features
Discover
Chapter 16: Q 3P (page 486)
How many products might be formed on chlorination of o-xylene (o-dimethylbenzene), m-xylene, and p-xylene?
 Chlorination of o-xylene
Chlorination of o-xylene
 Chlorination of meta-xylene
Chlorination of meta-xylene
 Chlorination of para-xylene
Chlorination of para-xylene
All the tools & learning materials you need for study success - in one app.
Get started for free
Phenylboronic acid, , is nitrated to give 15% ortho substitution product and 85% meta. Explain the meta-directing effect of the group.
Question: Predict the major products of the following reactions:
(a) Nitration of bromobenzene
(b) Bromination of nitrobenzene
(c) Chlorination of phenol
(d) Bromination of aniline
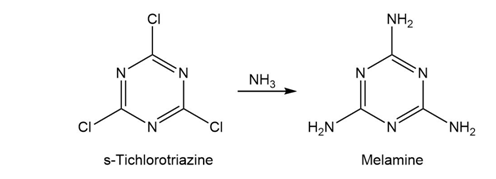
Melamine, used as a fire retardant and a component of the writing surface of white boards, can be prepared from s-trichlorotriazine through a series of SNAr reactions with ammonia. The first substitution takes place rapidly at room temperature. The second substitution takes place near 100 °C, and the third substitution requires even higher temperature and pressure. Provide an explanation for this reactivity.
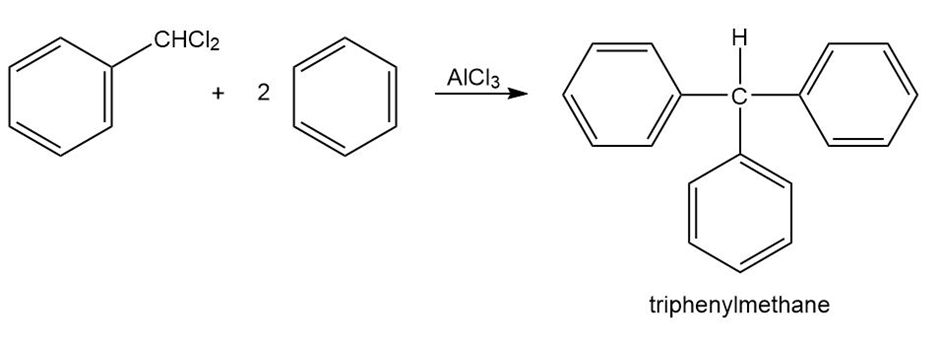
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of . Propose a mechanism for the reaction.
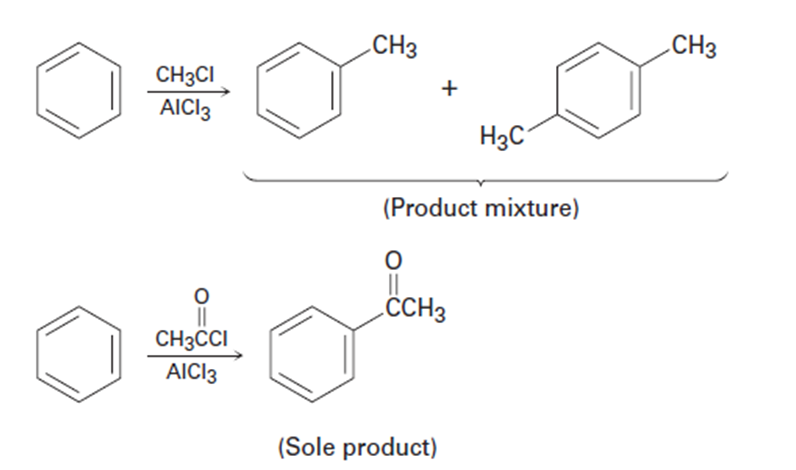
Use Figure 16-11 to explain why Friedel–Crafts alkylations often give polysubstitution but Friedel–Crafts acylations do not.
What do you think about this solution?
We value your feedback to improve our textbook solutions.