Chapter 16: Q33E (page 524)
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of . Propose a mechanism for the reaction.
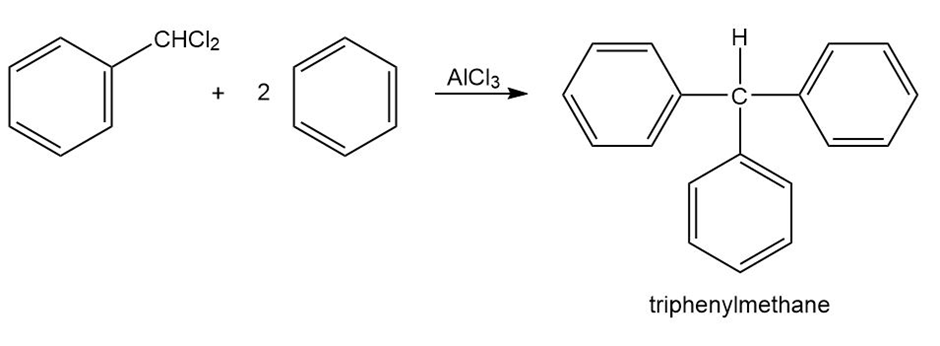
Short Answer
Mechanism followed as shown below.
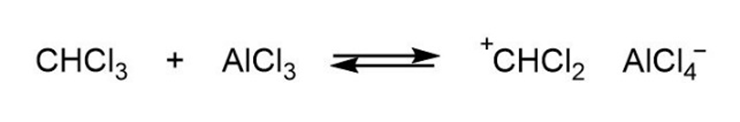
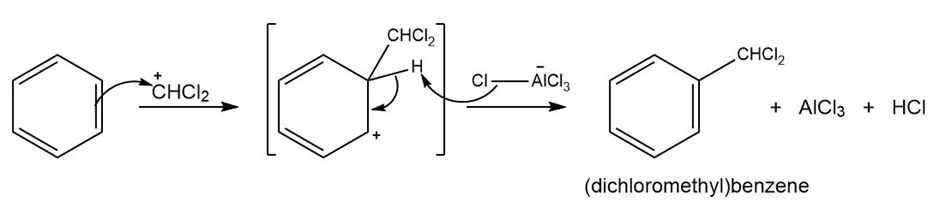
Learning Materials
Features
Discover
Chapter 16: Q33E (page 524)
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of . Propose a mechanism for the reaction.

Mechanism followed as shown below.


All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism to account for the reaction of benzene with 2,2,5,5-tetramethyltetrahydrofuran.
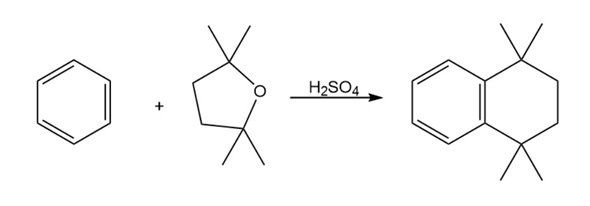
Refer to Table 6-3 on page 170 for a quantitative idea of the stability of a benzyl radical. How much more stable (in kJ/mol) is the benzyl radical than a primary alkyl radical? How does a benzyl radical compare in stability to an allylradical?
Would you expect p-methylphenol to be more acidic or less acidic than unsubstituted phenol? Explain. (See Problem 16-75.)
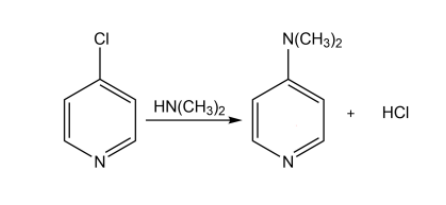
4- chloropyridine undergoes reaction with dimethylamine to yield 4-dimethylaminopyridine. Propose a mechanism for the reaction.
The carbocation electrophile in a Friedel–Crafts reaction can be generated by an alternate means than reaction of an alkyl chloride with. For example, reaction of benzene with 2-methylpropene in the presence of yields tert-butylbenzene. Propose a mechanism for this reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.