Chapter 16: Q70E (page 524)
Propose a mechanism to account for the reaction of benzene with 2,2,5,5-tetramethyltetrahydrofuran.
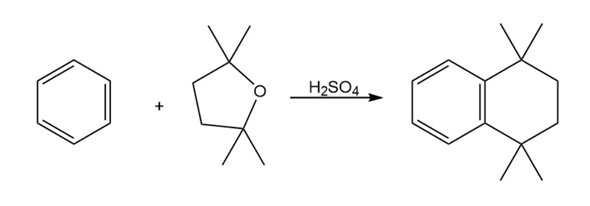
Short Answer
Mechanism followed as:
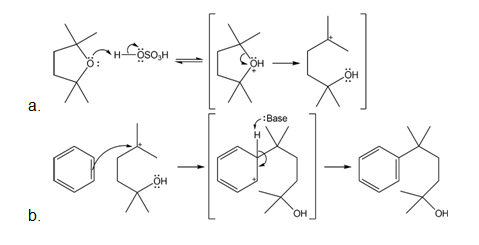
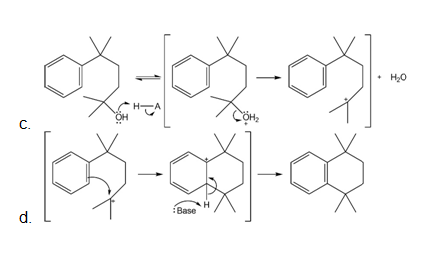
Learning Materials
Features
Discover
Chapter 16: Q70E (page 524)
Propose a mechanism to account for the reaction of benzene with 2,2,5,5-tetramethyltetrahydrofuran.

Mechanism followed as:


All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the electrophilic fluorination of benzene with.
Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.
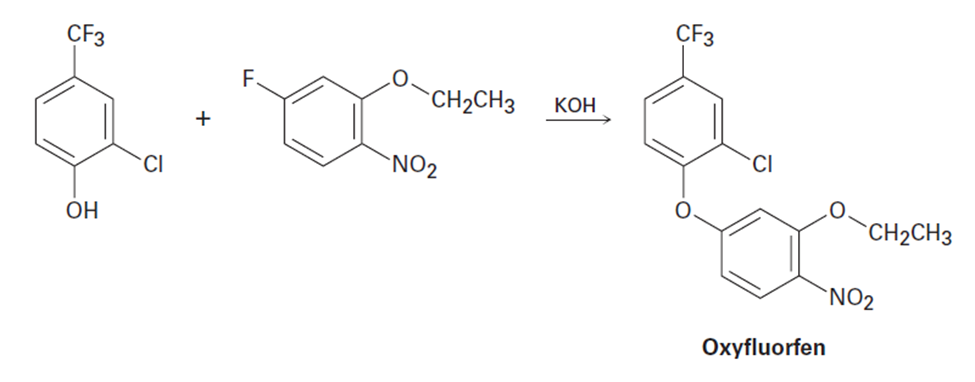
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose a mechanism.
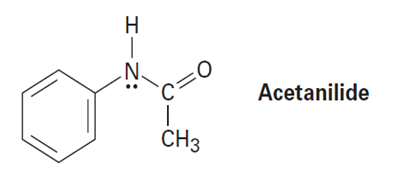
Acetanilide is less reactive than aniline toward electrophilic substitution. Explain
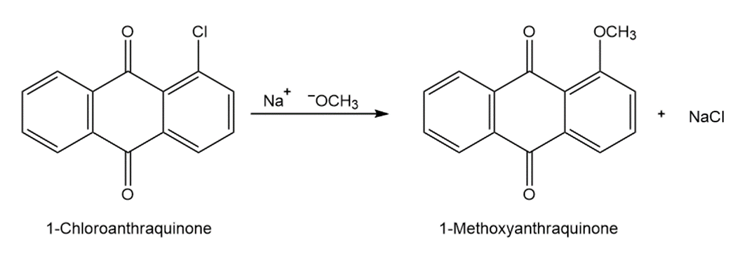
Propose a mechanism for the reaction of 1-chloroanthraquinone with methoxide ion to give the substitution product 1-methoxyanthraquinone. Use curved arrows to show the electron flow in each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.