Chapter 16: Q71E (page 524)
How would you synthesize the following compounds from benzene? Assume that ortho and para isomers can be separated.
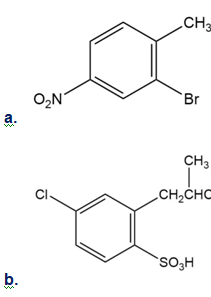
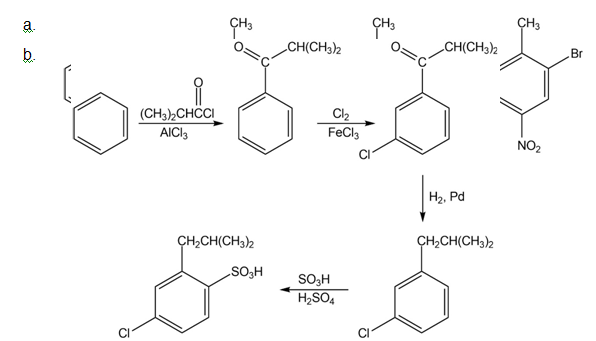
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q71E (page 524)
How would you synthesize the following compounds from benzene? Assume that ortho and para isomers can be separated.


All the tools & learning materials you need for study success - in one app.
Get started for free
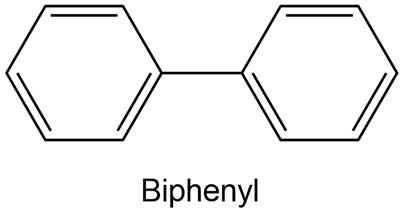
Using resonance structures of the intermediates, explain why bromination of biphenyl occurs at ortho and para positions rather than at meta.
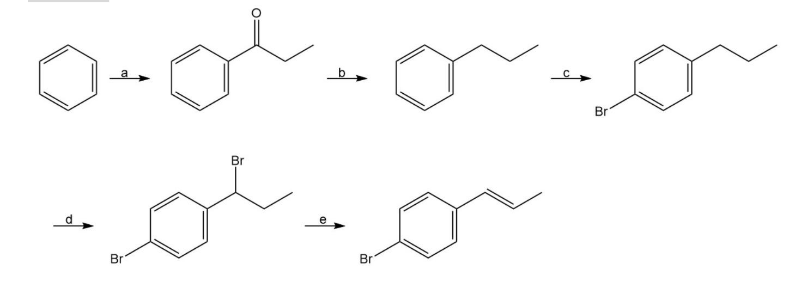
Question: Identify the reagents represented by the letters a–e in the following scheme:
Question: Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.
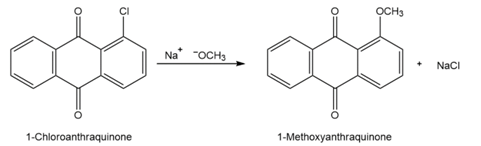
Propose a mechanism for the reaction of 1-chloroanthraquinone with methoxide ion to give the substitution product 1-methoxyanthraquinone. Use curved arrows to show the electron flow in each step.
Question: When benzene is treated with , deuterium slowly replaces all six hydrogens in the aromatic ring. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.