Chapter 16: Q 4P (page 486)
Question: When benzene is treated with , deuterium slowly replaces all six hydrogens in the aromatic ring. Explain.
Short Answer

Learning Materials
Features
Discover
Chapter 16: Q 4P (page 486)
Question: When benzene is treated with , deuterium slowly replaces all six hydrogens in the aromatic ring. Explain.

All the tools & learning materials you need for study success - in one app.
Get started for free
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose a mechanism.
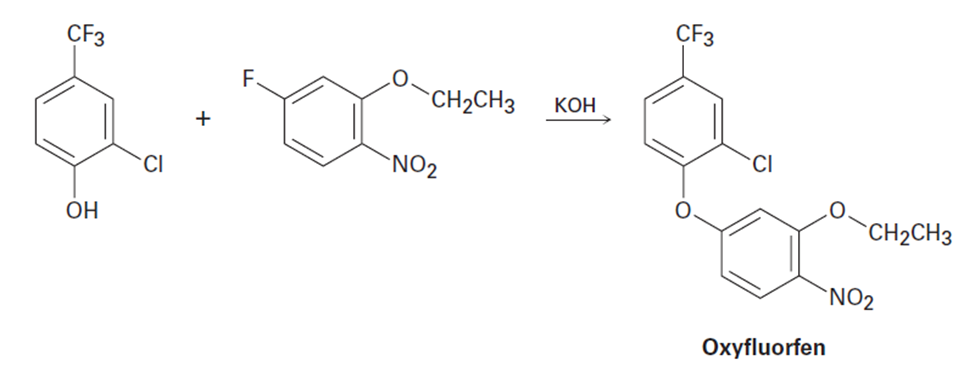
Question: You know the mechanism of HBr addition to alkenes, and you know the effects of various substituent groups on aromatic substitution. Use this knowledge to predict which of the following two alkenes reacts faster with HBr. Explain your answer by drawing resonance structures of the carbocation intermediates.
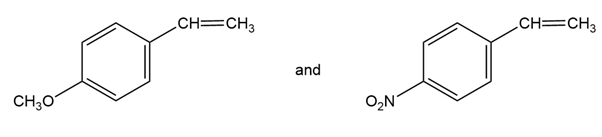
Acetanilide is less reactive than aniline toward electrophilic substitution. Explain
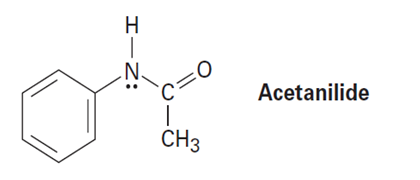
Would you expect p-methylphenol to be more acidic or less acidic than unsubstituted phenol? Explain. (See Problem 16-75.)
Name and draw the major product (s) of electrophilic chlorination of the following compounds:
What do you think about this solution?
We value your feedback to improve our textbook solutions.