Chapter 16: Q53E (page 524)
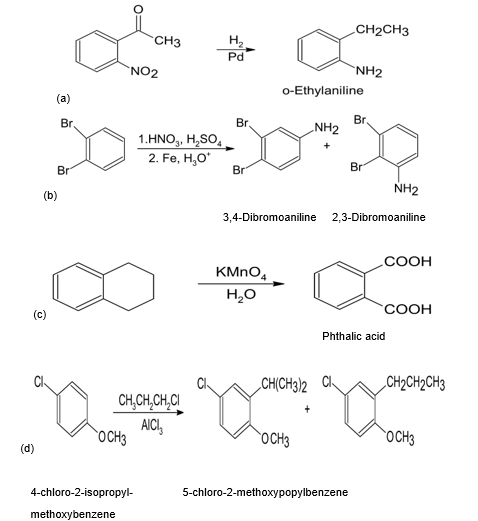
What product(s) would you expect to obtain from the following
reactions?
(a)
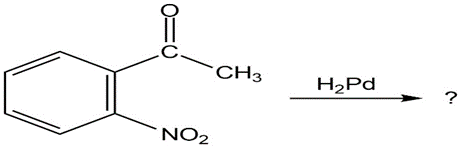
(b)
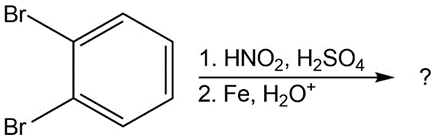
(c)
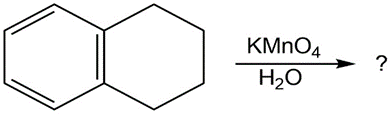
(d)
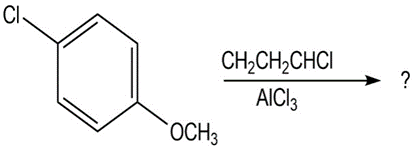
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 16: Q53E (page 524)
What product(s) would you expect to obtain from the following
reactions?
(a)

(b)

(c)

(d)

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
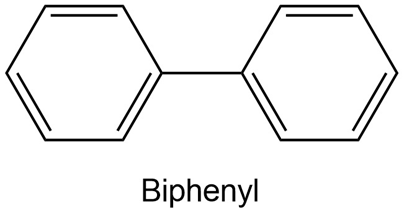
Using resonance structures of the intermediates, explain why bromination of biphenyl occurs at ortho and para positions rather than at meta.
Rank the following aromatic compounds in the expected order of their
reactivity toward Friedel–Crafts alkylation. Which compounds are
unreactive?
(a)Bromobenzene (b) Toluene (c) Phenol
(d)Aniline (e)Nitrobenzene (f)p-Bromotoluene
Draw resonance structures for the intermediates from the reaction of an electrophile at the ortho, meta, and para positions of nitrobenzene. Which intermediates are most stable?
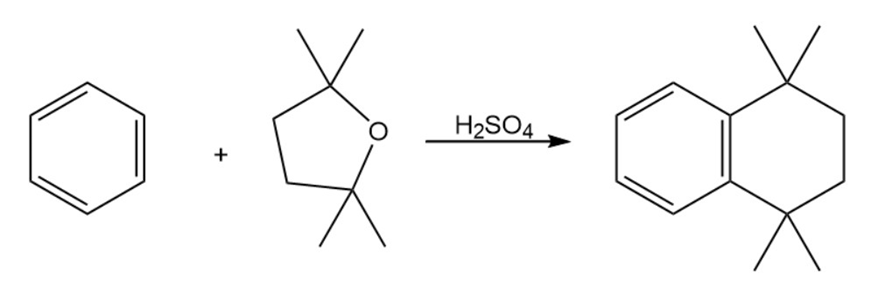
Propose a mechanism to account for the reaction of benzene with 2,2,5,5 tetramethyltetrahydrofuran.
What is the major monosubstitution product from the Friedel–Crafts reaction of benzene with 1-chloro-2-methylpropane in the presence of ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.